THE BREAST: Physiology & Lactation
Lactation is a highly coordinated physiological process that enables nourishment of the newborn through the production and delivery of breast milk. It involves complex interactions between mammary gland anatomy, hormonal signalling, neural reflexes and ongoing maternal–infant interaction.
Breast physiology begins long before birth, with mammary development occurring in puberty, maturing fully during pregnancy and becoming functionally active in the postpartum period. Understanding lactation is essential in healthcare, especially in maternal, neonatal and paediatric practice, as disruptions in this system can affect infant nutrition, maternal health and early bonding.
What You Need to Know
The breast is a hormonally responsive organ specialised for milk production and delivery after birth. Its functional units are the mammary glands, which are organised into lobes, lobules, and microscopic alveoli. The alveoli are lined with milk-producing epithelial cells and surrounded by myoepithelial cells, which contract to move milk into the ductal system and toward the nipple. This branching duct network allows milk to be transported efficiently from its site of synthesis to the infant.
During pregnancy, the breast undergoes marked structural and functional preparation for lactation. Estrogen stimulates ductal growth and branching, progesterone promotes development of lobules and alveoli, and prolactin primes the epithelial cells for milk synthesis. Although prolactin levels rise steadily throughout pregnancy, high circulating progesterone prevents large-scale milk secretion, ensuring that full lactation does not begin until after delivery.
Following birth, removal of the placenta leads to a rapid fall in progesterone and estrogen, triggering lactogenesis II, the onset of copious milk production. From this point onward, milk supply becomes primarily regulated by neuroendocrine reflexes linked to infant feeding. Key hormonal mechanisms involved in lactation include:
Prolactin, which stimulates milk synthesis within the alveolar cells
Oxytocin, which causes contraction of myoepithelial cells and milk ejection during the let-down reflex
Neural feedback from suckling, which maintains ongoing hormone release and milk production
Effective and frequent infant suckling is essential for sustaining lactation. Sensory input from the nipple stimulates continued prolactin and oxytocin release, reinforcing milk synthesis and delivery. This demand-driven system allows milk supply to adapt to the infant’s needs and highlights the close integration between breast physiology, hormonal regulation, and feeding behaviour.
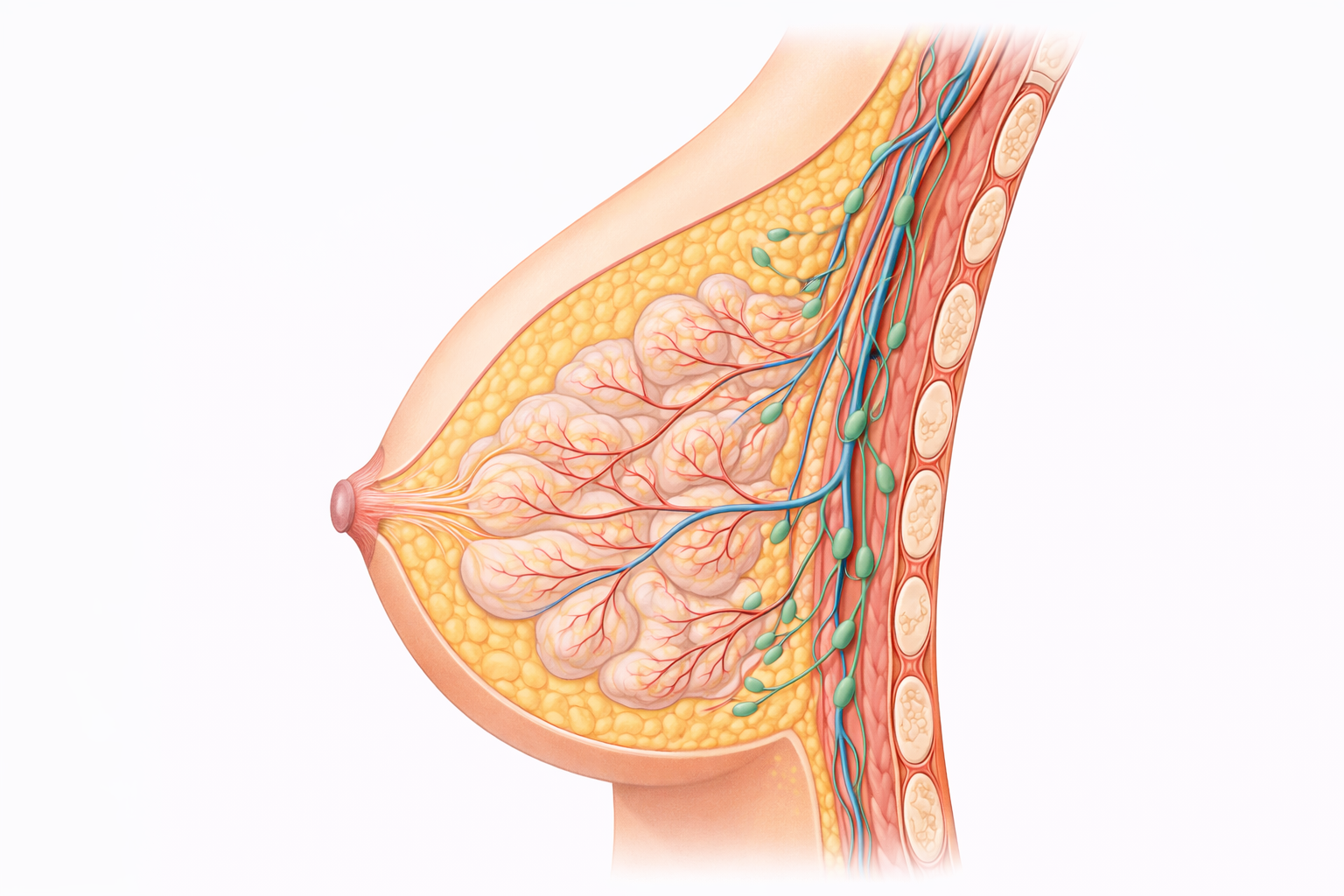
Image: The breast is composed of glandular lobules that produce milk, connected by ducts that converge at the nipple. These structures are embedded within adipose (fat) tissue, which gives the breast its shape, while underlying the breast are the pectoral muscles and rib cage. Lymphatic vessels and nodes are also present, playing a key role in fluid drainage and immune function.
Beyond the Basics
Mammary Anatomy and Functional Microstructure
The breast is organised into 15 to 20 lobes arranged radially around the nipple, each draining through its own ductal pathway. Within each lobe are smaller lobules composed of clusters of alveoli, the primary sites of milk synthesis. Alveoli are lined by secretory epithelial cells responsible for producing milk constituents and are encircled by myoepithelial cells that provide the contractile force required for milk movement.
Milk produced within the alveoli passes into progressively larger ducts that converge toward the nipple, where multiple duct openings allow milk to exit during feeding. Structural support is provided by fibrous connective tissue known as Cooper’s ligaments, which help maintain breast shape. Adipose tissue makes up the majority of visible breast volume and varies widely between individuals, but it does not contribute to milk production. Functional capacity is determined by glandular tissue rather than breast size.
Hormonal Preparation During Pregnancy
Pregnancy initiates extensive mammary development in preparation for lactation. Estrogen stimulates elongation and branching of the ductal system, increasing the capacity to transport milk. Progesterone promotes growth and differentiation of lobules and alveoli, establishing the architecture required for milk synthesis. Prolactin, secreted by the anterior pituitary, primes secretory epithelial cells by activating the cellular machinery needed for lactogenesis.
Placental hormones, including human placental lactogen, further enhance mammary growth and metabolic readiness. Despite high prolactin levels during pregnancy, sustained estrogen and progesterone concentrations inhibit copious milk production. This ensures that full lactation does not begin until after birth, when nutrient transfer to the infant becomes necessary.
Lactogenesis II and Early Milk Production
Following delivery of the placenta, circulating progesterone levels fall rapidly. This hormonal shift removes the inhibitory effect on prolactin, triggering lactogenesis II, the onset of abundant milk production. This transition typically occurs within two to four days postpartum and is often perceived as breast fullness or engorgement.
The earliest milk, colostrum, is produced in small volumes but is highly concentrated with immunological components. It contains secretory immunoglobulin A, lactoferrin, leukocytes, and growth factors that provide mucosal protection and support early gut development. As milk volume increases, colostrum gradually transitions to mature milk, with changes in macronutrient composition that align with the infant’s evolving nutritional needs.
Prolactin and Regulation of Milk Synthesis
Prolactin is the primary hormone responsible for ongoing milk synthesis. It acts directly on secretory epithelial cells to stimulate transcription of milk protein genes and regulate lactose, fat, and protein production. Prolactin secretion rises in response to nipple stimulation, with particularly strong nocturnal release, reinforcing the importance of frequent and effective feeding in the early postpartum period.
In addition to its role in lactation, prolactin suppresses hypothalamic gonadotrophin-releasing hormone, reducing luteinising hormone and follicle-stimulating hormone secretion. This mechanism underlies lactational amenorrhoea and contributes to temporary suppression of ovulation during exclusive breastfeeding.
Oxytocin and Milk Ejection
Oxytocin governs milk ejection rather than milk production. Released from the posterior pituitary in response to nipple stimulation, oxytocin causes myoepithelial cells surrounding the alveoli to contract, forcing milk into the ductal system and toward the nipple. This coordinated contraction produces the let-down reflex that allows milk to be available to the infant.
Oxytocin release is sensitive to emotional and environmental factors. Auditory cues such as an infant’s cry or anticipation of feeding can trigger let-down, while stress, pain, or anxiety may inhibit oxytocin release and delay milk ejection despite adequate milk synthesis. This distinction explains why perceived low milk supply may sometimes reflect impaired let-down rather than reduced production.
Maintenance of Milk Supply and Local Regulation
Long-term milk production is regulated by demand and milk removal rather than baseline hormone levels. Effective and frequent emptying of the breast increases prolactin receptor expression on secretory cells and supports continued synthesis. Local regulatory mechanisms also play a role, particularly the feedback inhibitor of lactation, a whey protein that accumulates when milk is not removed.
When milk remains in the breast, rising concentrations of this inhibitor downregulate milk synthesis. Conversely, frequent suckling or expression reduces local inhibition and promotes ongoing production. This adaptive system allows milk supply to match infant needs over time rather than remaining fixed.
Milk Composition and Functional Adaptability
Breast milk is a dynamic biological fluid that changes within a single feed, across the day, and over the course of lactation. Early milk within a feed is relatively lower in fat and higher in lactose, supporting hydration and energy delivery, while milk later in the feed contains higher fat concentrations that promote satiety and growth.
Beyond macronutrients, breast milk contains immunological and bioactive components that support infant health. Antibodies, antimicrobial peptides, and immune cells protect against infection, while hormones such as leptin, adiponectin, and growth factors influence appetite regulation, metabolic programming, and tissue development. This capacity for ongoing adaptation highlights lactation as an active, responsive physiological process rather than a static mode of nutrition delivery.
Clinical Connections
Breast physiology explains many of the changes and symptoms seen across the lifespan, not only during lactation. Because breast tissue is highly responsive to hormonal signals, fluctuations in estrogen, progesterone, prolactin, and oxytocin influence breast size, texture, sensitivity, and function from puberty through menopause. Cyclical tenderness, diffuse nodularity, and transient fullness are common and usually reflect normal endocrine variation rather than disease.
During pregnancy and the postpartum period, physiological changes become more pronounced as glandular tissue expands, vascularity increases, and milk synthesis begins. When milk removal is ineffective or hormonal transitions are disrupted, breast discomfort and inflammation may develop. Poor latch, infrequent feeding, or impaired milk ejection can lead to engorgement, ductal obstruction, and mastitis. Stress, pain, and fatigue may inhibit oxytocin release, limiting milk flow even when milk production is adequate. Structural factors such as inverted nipples, focal duct obstruction, or retained placental tissue can further complicate this process, while endocrine disorders affecting prolactin may alter milk synthesis.
Across clinical contexts, many common breast presentations can be understood by linking symptoms back to tissue structure, milk dynamics, and hormonal regulation:
Cyclical mastalgia or nodularity, driven by estrogen and progesterone effects on ductal and stromal tissue
Engorgement, blocked ducts, or mastitis, resulting from milk stasis and local inflammatory responses
Nipple pain or trauma, usually mechanical in origin and related to tissue stress or latch dynamics
Perceived low milk supply, often reflecting impaired milk transfer rather than reduced glandular function
Knowledge of normal breast physiology is also critical when assessing possible pathology. Hormone-related breast changes tend to be bilateral, diffuse, and cyclical, whereas concerning features are more likely to be focal, progressive, or persistent. New breast lumps, areas of focal thickening, skin dimpling, nipple inversion, or spontaneous nipple discharge that do not resolve with hormonal fluctuation require assessment. Recognising an individual’s baseline breast pattern supports earlier detection of abnormal change while avoiding unnecessary investigation of normal variation.
Endocrine influences on the breast extend beyond lactation. Prolactin-mediated suppression of gonadotrophin release explains lactational amenorrhoea and has implications for fertility counselling, as ovulation can resume before menstruation returns. Later in life, declining estrogen contributes to breast involution and altered tissue composition, which should be considered when interpreting symptoms or imaging findings.
Concept Check
How do estrogen and progesterone prepare the breast for lactation during pregnancy?
Why does milk production increase only after delivery of the placenta?
What roles do prolactin and oxytocin play in lactation, and how do they differ?
How does the frequency of infant feeding influence ongoing milk supply?
What are some physiological reasons why milk ejection may be impaired even when milk supply is adequate?