THE UTERUS: Structure & Function
The uterus is a central organ of the female reproductive system, responsible for supporting implantation, sustaining pregnancy and facilitating childbirth. Its structure is uniquely adapted to undergo profound cyclic and gestational changes, shifting from a small pelvic organ to an expanded structure capable of supporting a developing fetus. Beyond pregnancy, the uterus plays a critical role in menstrual physiology through coordinated interactions between its muscular, vascular and epithelial layers.
What You Need to Know
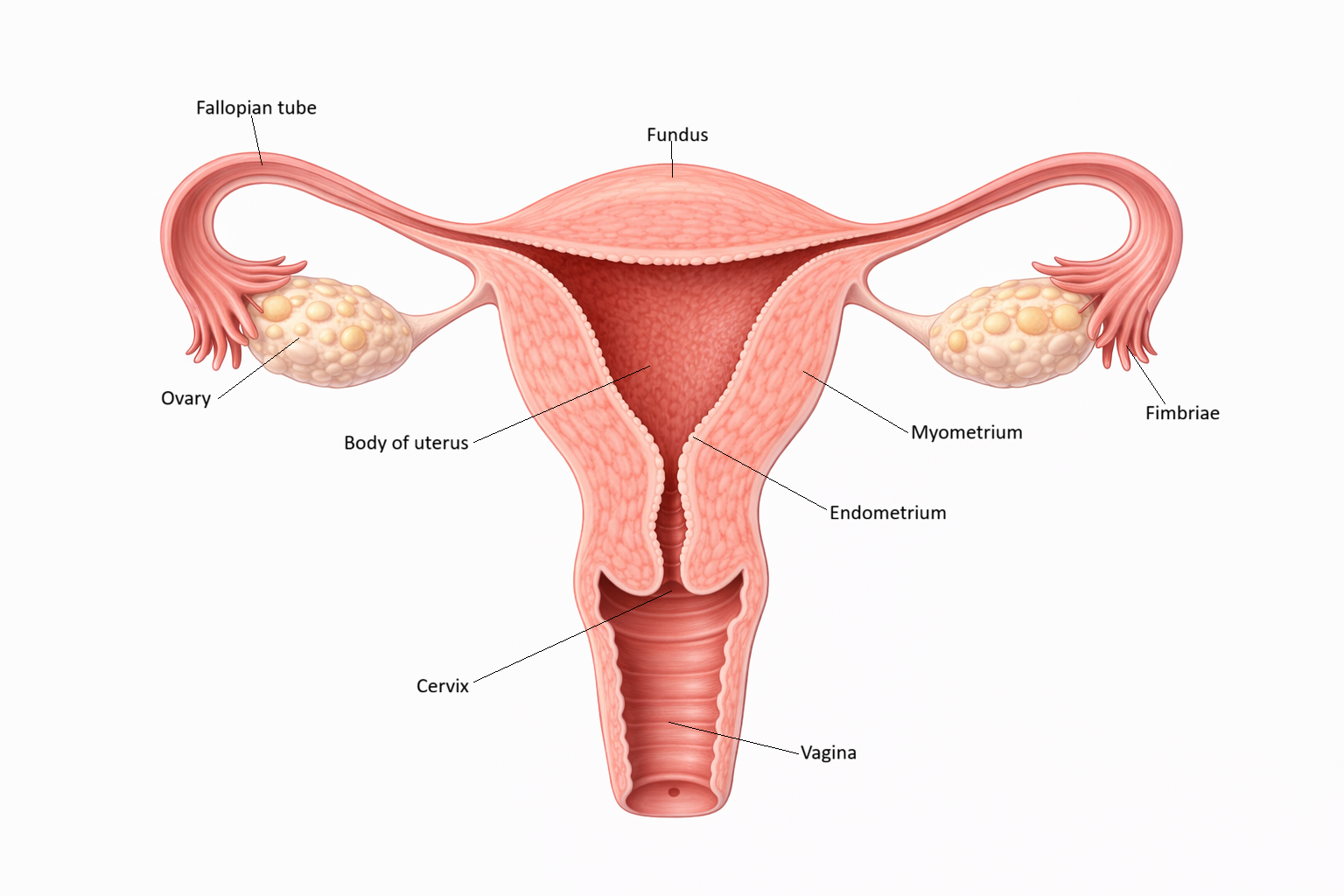
The uterus is a highly specialised muscular organ designed to support implantation, pregnancy, and childbirth. It is positioned within the pelvis between the bladder anteriorly and the rectum posteriorly, allowing close anatomical and functional relationships with surrounding pelvic structures. Anatomically, the uterus is divided into the fundus, which forms the rounded superior portion, the body or corpus, which makes up the majority of the uterine mass, and the cervix, which projects into the vagina and forms the gateway between the uterine cavity and the lower reproductive tract.
The uterine wall is composed of three distinct layers, each contributing to uterine function in different physiological states. The endometrium is the innermost layer and undergoes cyclical changes in response to ovarian hormones. Beneath this lies the myometrium, a thick layer of interlacing smooth muscle fibres responsible for uterine contractility. The outermost layer, the perimetrium, is a serosal covering that provides structural protection and reduces friction with surrounding pelvic organs.
Structural organisation allows the uterus to respond dynamically to changing hormonal environments across the lifespan. Key functional features of the uterine layers include:
Endometrium, which regenerates and differentiates each cycle to support implantation and early pregnancy
Myometrium, which generates coordinated contractions during menstruation and labour
Perimetrium, which provides support and forms part of the pelvic peritoneal surface
Hormonal regulation drives marked changes in uterine structure over time. Estrogen and progesterone orchestrate cyclical endometrial growth, differentiation, and shedding during the menstrual cycle. During pregnancy, rising hormone levels stimulate smooth muscle hypertrophy, increased uterine blood flow, and extensive vascular remodelling to support the developing fetus. Following childbirth, withdrawal of pregnancy hormones allows uterine involution, returning the uterus to its non-pregnant size and function. This capacity for repeated structural transformation highlights the uterus as one of the most adaptable organs in human physiology.
Image: The uterus is a hollow muscular organ composed of a thick outer myometrium and an inner endometrium, which lines the uterine cavity. The endometrium undergoes cyclical changes in response to hormones, preparing for potential implantation, while the fallopian tubes extend laterally toward the ovaries to facilitate the transport of the oocyte.
Beyond the Basics
Gross Anatomy and Spatial Relationships
The uterus is typically anteverted and anteflexed, meaning it tilts forward over the bladder and bends slightly at the junction between the cervix and body. This orientation supports efficient load distribution within the pelvis and accommodates changes in uterine size across the menstrual cycle and pregnancy. A network of ligaments provides stability without rigid fixation. The broad ligament acts as a peritoneal fold supporting the uterus laterally, while the uterosacral and round ligaments help maintain position and limit excessive displacement as uterine volume increases.
Vascular supply to the uterus is extensive and highly adaptable. The uterine arteries, branches of the internal iliac arteries, form rich anastomoses that ensure consistent perfusion of the endometrium, myometrium, and cervix even as tissue demands fluctuate. Venous drainage occurs through a plexiform network that expands markedly during pregnancy to accommodate increased blood flow. The uterus also maintains close functional relationships with the pelvic floor musculature, which contributes to organ support, continence, and the biomechanics of labour.
Endometrium: Cyclical Transformation and Hormone Responsiveness
The endometrium is a hormonally responsive mucosal lining composed of glandular epithelium, connective tissue stroma, and a dense vascular network. It is organised into two layers with distinct roles across the cycle. The basal layer remains intact and serves as the regenerative source, while the functional layer undergoes cyclical growth, differentiation, and shedding.
During the proliferative phase, estrogen stimulates rapid cellular division within glands and stroma, leading to thickening of the functional layer and elongation of spiral arteries. After ovulation, progesterone induces secretory transformation, with glands becoming coiled and producing nutrient-rich secretions that support early embryonic development. In the absence of implantation, withdrawal of progesterone leads to constriction of spiral arteries, localised ischemia, and breakdown of the functional layer, resulting in menstruation. This tightly regulated vascular response is central to normal cycle timing and bleeding patterns.
Myometrium: Contractility and Structural Adaptation
The myometrium is a thick, interwoven layer of smooth muscle fibres supported by connective tissue and an extensive vascular network. It exhibits remarkable plasticity, particularly during pregnancy, when smooth muscle cells enlarge rather than multiply, allowing the uterus to expand substantially without compromising integrity. Outside pregnancy, the myometrium generates low-amplitude, rhythmic contractions that assist menstrual shedding and contribute to uterine tone.
As term approaches, myometrial behaviour changes in preparation for labour. Increased oxytocin receptor expression, enhanced gap junction formation, and altered ion channel activity improve electrical coupling between muscle cells. This coordinated activity enables powerful, synchronised contractions that facilitate cervical dilation and fetal expulsion. After delivery, sustained myometrial contraction compresses uterine blood vessels at the placental site, a critical mechanism for limiting postpartum blood loss.
Perimetrium: Serosal Interface and Protection
The perimetrium is the thin serosal covering of the uterus, derived from visceral peritoneum. Its primary role is to provide a smooth, lubricated surface that allows the uterus to move freely against adjacent pelvic organs as size and position change. Although it contributes little to contractile or endocrine function, the perimetrium maintains structural continuity with surrounding peritoneal surfaces and forms an important anatomical boundary within the pelvic cavity.
Cervical Structure and Functional Integration
The cervix, while anatomically distinct, is integral to overall uterine physiology. Its dense collagen-rich composition allows it to remain firm and closed through most of the cycle and throughout pregnancy, providing mechanical support and a protective barrier. Cervical mucus properties vary markedly in response to hormonal shifts. Under estrogen influence, mucus becomes thin and hydrated, facilitating sperm passage into the uterine cavity. Under progesterone dominance, it thickens and becomes less permeable, limiting sperm entry and reducing pathogen ascent.
As labour approaches, cervical tissue undergoes extensive remodelling driven by prostaglandins and inflammatory mediators. Collagen fibres reorganise, water content increases, and the cervix softens and dilates, allowing effective transmission of myometrial forces during childbirth. This coordinated cervical response highlights how uterine function depends not only on muscle contraction but also on precise structural and biochemical adaptation.
Clinical Connections
Disruption of normal uterine structure or hormone responsiveness can affect fertility, bleeding patterns, and pregnancy outcomes. Because the uterus is highly sensitive to estrogen and progesterone, even subtle endocrine imbalance can alter endometrial development or myometrial behaviour. Clinically, this often presents as abnormal uterine bleeding, pain, or difficulty achieving or maintaining a pregnancy rather than isolated uterine pathology.
Several common gynaecological conditions reflect altered uterine architecture or function. Endometriosis and adenomyosis change the normal relationship between endometrial tissue and the myometrium, contributing to chronic pelvic pain, dysmenorrhoea, and reduced fertility. Uterine fibroids, or leiomyomas, arise from the myometrium and may distort the uterine cavity, impair implantation, or increase menstrual blood loss depending on their size and location. Endometrial hyperplasia and carcinoma are most often linked to prolonged unopposed estrogen exposure, where the absence of progesterone-driven differentiation allows excessive endometrial proliferation. Key clinical consequences of uterine dysfunction include:
Abnormal uterine bleeding, often related to anovulation, fibroids, or endometrial pathology
Infertility or implantation failure, due to impaired endometrial receptivity or cavity distortion
Pelvic pain and pressure symptoms, reflecting altered myometrial or endometrial structure
During pregnancy, uterine physiology remains central to maternal and fetal health. Adequate remodelling of uterine spiral arteries is essential for placental perfusion, and failure of this process contributes to hypertensive disorders such as pre-eclampsia. Structural and connective tissue integrity is also important beyond pregnancy. Weakness of pelvic floor muscles or ligamentous support can lead to uterine prolapse, particularly after vaginal birth or with increasing age, affecting continence, comfort, and quality of life.
Understanding normal uterine function also explains the mechanisms of widely used contraceptive methods. Hormonal contraception suppresses ovulation and limits endometrial proliferation, creating an environment that is unreceptive to implantation. Intrauterine devices act locally within the uterine cavity, altering the inflammatory and biochemical environment to reduce fertilisation and implantation. In clinical practice, linking symptoms and treatment effects back to uterine physiology allows more precise assessment of bleeding disorders, fertility concerns, and reproductive health across the lifespan.
Concept Check
How do the endometrial layers differ in structure and function?
What hormonal changes drive the transition from the proliferative to the secretory phase of the endometrium?
How does the myometrium adapt structurally during pregnancy and labour?
Why are spiral arteries essential for both normal menstruation and early pregnancy?
How does cervical mucus change across the menstrual cycle, and what is the physiological purpose of these changes?