Alveolar Structure
The alveoli are the fundamental functional units of the respiratory system, responsible for the exchange of oxygen and carbon dioxide that sustains cellular metabolism. Each lung contains millions of alveoli, forming an extensive surface area optimised for gas exchange. These delicate structures must remain open, thin, moist, and well-perfused to function effectively. Any disruption to their architecture, whether through inflammation, collapse, or destruction, has profound consequences for oxygenation and respiratory function.
What You Need to Know
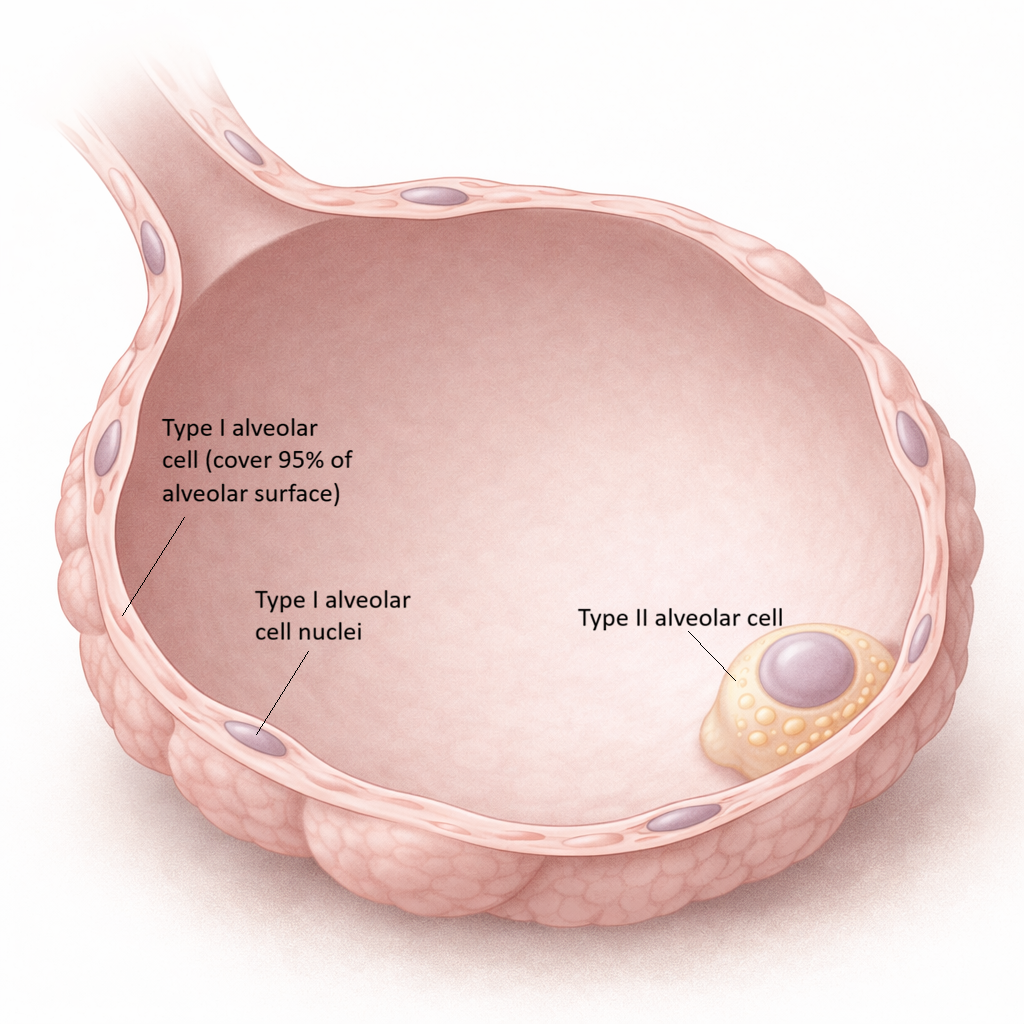
Alveoli are tiny sac-like structures lined predominantly by Type I alveolar cells, which form a thin, flat epithelial layer optimised for diffusion. These cells cover approximately 95% of the alveolar surface and create a minimal barrier for gas movement between the alveolar air and capillary blood. Interspersed between them are Type II alveolar cells, which are more cuboidal in shape and far more metabolically active.
Key components of alveoli:
Type I alveolar cells → thin, flat cells forming the diffusion surface
Type II alveolar cells → surfactant production and alveolar stability
Alveolar macrophages → clearance of inhaled particles and pathogens
Their primary function is to synthesise and secrete pulmonary surfactant, a phospholipid-rich substance that reduces surface tension and prevents alveolar collapse, especially during expiration. Without adequate surfactant, the alveoli would constantly deflate, increasing the work of breathing and impairing oxygenation.
The alveolar space also contains resident macrophages, which represent the final line of defence against inhaled pathogens and debris. These macrophages patrol the alveolar surface, clearing dust, bacteria, and other foreign particles before they can trigger widespread inflammation. Surrounding each alveolus is an extensive network of capillaries. The thin respiratory membrane, composed of Type I alveolar cells, a fused basement membrane, and capillary endothelial cells, allows rapid diffusion of gases between air and blood.
Image: cross section of an alveolus
Beyond the Basics
Alveolar Structure and Elastic Recoil
The structure of the alveoli is precisely adapted to their function in gas exchange. Their near-spherical shape maximises surface area relative to volume, allowing millions of alveoli to collectively provide an enormous interface for oxygen and carbon dioxide diffusion. This geometric efficiency is matched by the elastic properties of the alveolar walls, which enable alveoli to expand during inspiration and recoil passively during expiration.
Elastic recoil is generated by a network of elastin and collagen fibres embedded within the alveolar septa. These fibres store energy during lung inflation and release it during expiration, reducing the muscular effort required to breathe out. Loss of this elastic framework, as seen in emphysema, fundamentally alters alveolar mechanics. Destruction of alveolar walls not only reduces the available surface area for gas exchange but also diminishes elastic recoil, leading to airway collapse during expiration, air trapping, and progressive breathlessness.
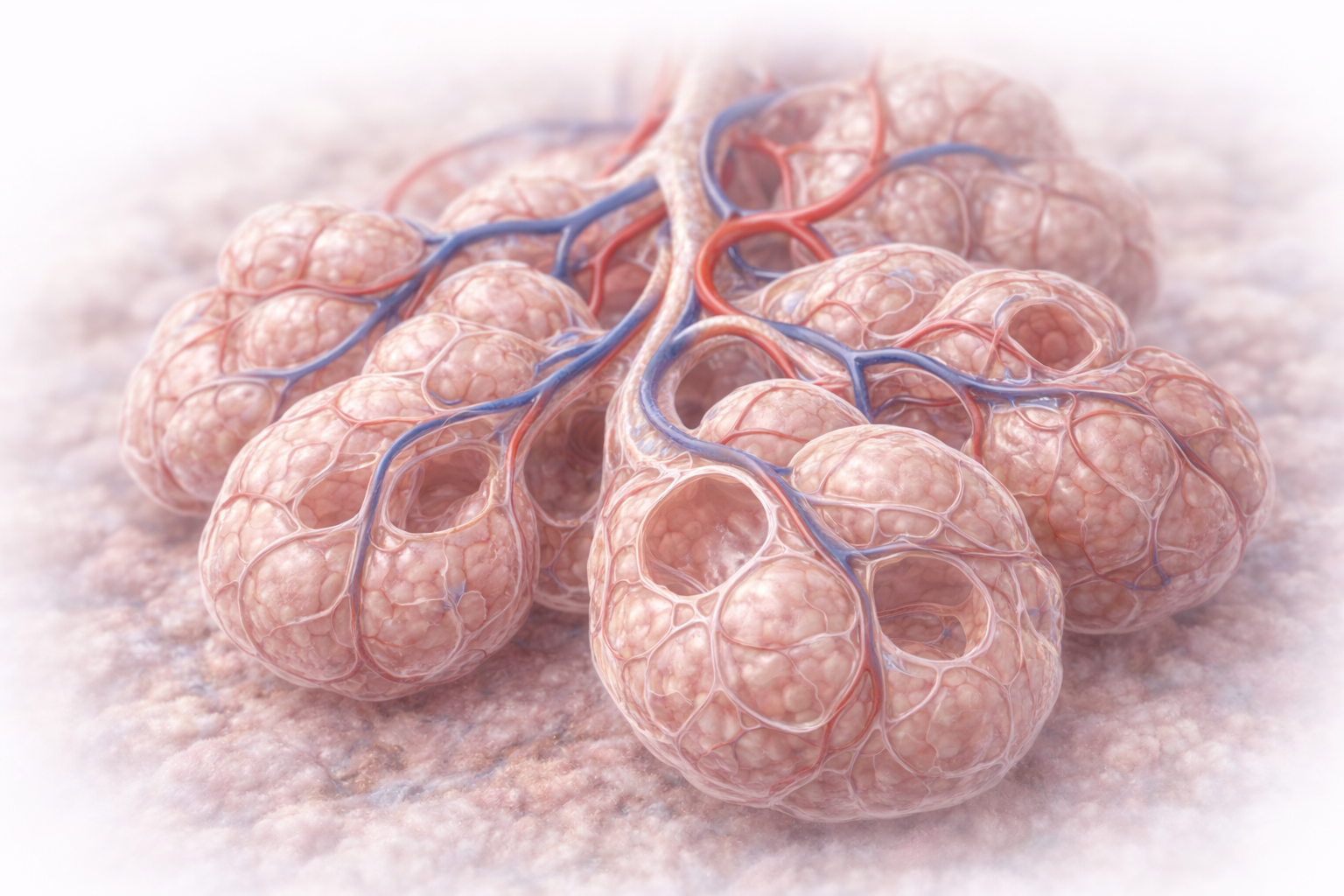
Image: Cluster of alveoli showing their thin, delicate walls and close association with surrounding capillaries. The minimal thickness of the alveolar–capillary membrane allows rapid gas exchange between air and blood, while the extensive surface area maximises efficiency.
Surfactant and Alveolar Stability
Pulmonary surfactant is essential for maintaining alveolar stability across the wide range of lung volumes encountered during normal breathing. By reducing surface tension at the air–liquid interface, surfactant lowers the pressure required to inflate alveoli and prevents collapse at end-expiration.
The importance of surfactant becomes clear when considering the Law of Laplace, which states that the pressure required to keep a spherical structure open is directly proportional to surface tension and inversely proportional to radius. Without surfactant, smaller alveoli would experience higher internal pressures than larger alveoli and would therefore tend to collapse into them. Surfactant counteracts this tendency by reducing surface tension more effectively in smaller alveoli, equalising pressures and allowing alveoli of different sizes to coexist stably.
This mechanism is critically important in newborns. In premature infants, immature Type II pneumocytes produce insufficient surfactant, resulting in widespread alveolar collapse, reduced lung compliance, and severe respiratory distress. Neonatal respiratory distress syndrome illustrates how disruption of surfactant production compromises both ventilation and gas exchange.
Alveolar Interdependence and Mechanical Support
Alveoli do not function as isolated units; instead, they are mechanically interconnected through shared septal walls and connective tissue. This alveolar interdependence means that the expansion of one alveolus helps support the patency of its neighbours, contributing to uniform lung inflation and overall mechanical stability.
When a cluster of alveoli collapses, as occurs in atelectasis, this supportive network is disrupted. The surrounding alveoli are subjected to increased mechanical stress and must generate greater forces to remain open. This redistribution of stress increases the likelihood of further collapse, creating a self-propagating cycle that reduces ventilated lung volume and impairs gas exchange.
Alveolar interdependence therefore plays a key role in maintaining lung stability under normal conditions, while its disruption explains why even small areas of collapse can have disproportionately large effects on ventilation and oxygenation.
Integration of Structure and Function
Efficient alveolar function depends on the seamless integration of surface area, elastic recoil, surfactant activity, and mechanical interdependence. Alterations in any one of these components can destabilise the alveolar network and compromise both ventilation and diffusion.
Understanding this tight coupling between structure and function provides a foundation for interpreting common respiratory pathologies and explains why diseases that disrupt alveolar integrity often produce profound and persistent respiratory impairment.
Clinical Connections
Diseases that alter alveolar structure often present with profound clinical consequences. Pneumonia fills the alveoli with inflammatory exudate, preventing ventilation and severely impairing gas exchange. Pulmonary oedema allows fluid to accumulate within the interstitial space and alveoli, increasing diffusion distance and reducing oxygen transfer. In ARDS, widespread inflammation injures both Type I and Type II alveolar cells, leading to a loss of surfactant, alveolar collapse, and stiff, non-compliant lungs that are extremely difficult to ventilate.
Common mechanisms of alveolar dysfunction:
Alveolar filling → impaired ventilation (pneumonia, pulmonary oedema)
Barrier thickening → reduced diffusion (oedema, fibrosis)
Surface area loss → impaired gas exchange (emphysema)
Surfactant loss → alveolar collapse (ARDS)
Emphysema demonstrates the opposite problem: destruction rather than filling. Loss of alveolar walls reduces surface area, damages capillaries, and impairs diffusion. The lung becomes hyperinflated, yet gas exchange worsens. Meanwhile, conditions such as pulmonary fibrosis thicken the respiratory membrane, slowing diffusion and causing exertional oxygen desaturation long before resting oxygen levels fall.
The health of the alveoli is therefore central to overall respiratory function, explaining why many respiratory diseases, despite differing causes, ultimately manifest as hypoxaemia and breathlessness.
Concept Check
Why are Type II alveolar cells essential for maintaining alveolar stability?
How does surfactant help prevent alveolar collapse?
Why does emphysema reduce the efficiency of gas exchange?
How does pneumonia interfere with the normal function of the alveoli?
Why does pulmonary fibrosis cause early exertional breathlessness?