Respiratory Defence Mechanisms
The respiratory system is constantly exposed to the external environment, inhaling millions of particles, pathogens, and chemical irritants every day. Despite this exposure, the lungs remain remarkably sterile under normal conditions, thanks to a sophisticated network of defence mechanisms. These defences begin in the upper airway and extend deep into the alveoli, working together to filter air, trap harmful substances, neutralise pathogens, and remove debris. Understanding these mechanisms explains why certain diseases impair lung defence and why others cause exaggerated inflammatory responses.
What You Need to Know
The respiratory system is continuously exposed to microorganisms, dust, smoke, and environmental toxins, so it relies on a layered defence system that begins in the upper airways and becomes increasingly specialised as air moves deeper into the lungs. The nasal cavity acts as the first physical filter. Nasal hairs and the complex, curved surfaces of the turbinates (conchae) create turbulent airflow, causing large particles to collide with the moist mucosal lining rather than travelling into the lower airways. At the same time, the nasal mucosa warms and humidifies incoming air, protecting delicate airway tissues and maintaining optimal conditions for gas exchange.
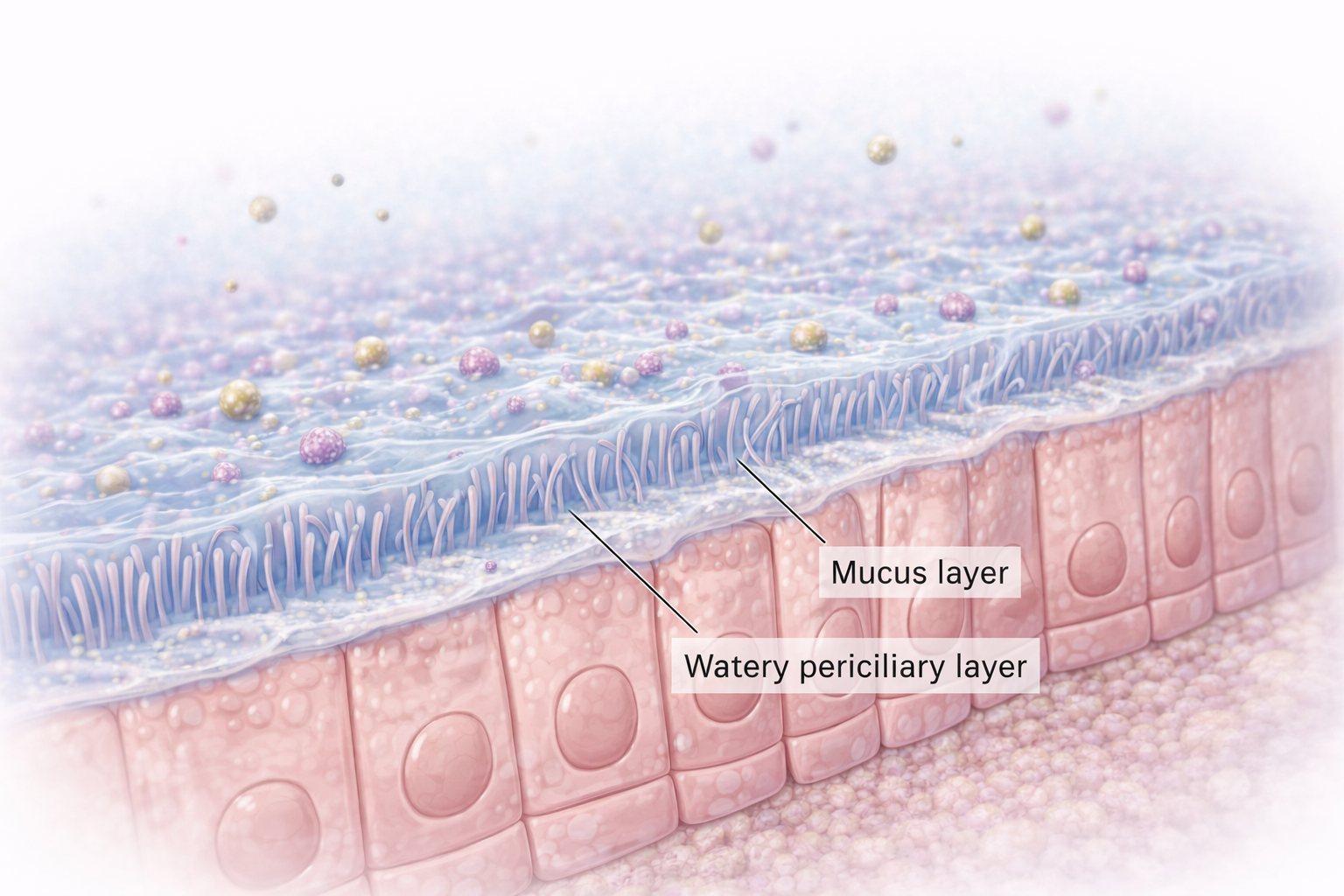
In the trachea and bronchi, protection is dominated by the mucociliary escalator - a coordinated clearance system in which sticky mucus traps inhaled particles and microorganisms, while millions of microscopic cilia beat rhythmically upward, moving the mucus toward the throat. Once there, it can be swallowed and destroyed by gastric acid or expelled by coughing. This mechanism prevents contaminants from reaching the fragile alveoli and is one of the most important everyday defence systems in the lungs.
Several structural and cellular features work together to keep the conducting airways sterile:
Goblet cells and submucosal glands produce mucus that traps pathogens
Ciliated epithelial cells move this mucus upward toward the pharynx
Cough and sneeze reflexes provide rapid clearance when irritation is detected
Once air reaches the alveoli, mechanical defences are largely absent, so immune defences take over. Alveolar macrophages act as the lungs’ resident immune sentinels. They continuously patrol the alveolar surfaces, engulfing bacteria, viruses, fungal spores, and particulate matter that bypass the upper airway defences. After ingesting a threat, macrophages either destroy it directly or signal for reinforcement by releasing cytokines (chemical messengers) that recruit neutrophils and other immune cells from the bloodstream.
Pulmonary surfactant also contributes to defence. In addition to preventing alveolar collapse, surfactant contains proteins with antimicrobial properties that interfere with bacterial membranes and viral attachment. Together, macrophages, surfactant, and recruited immune cells form a highly effective barrier against infection while minimising unnecessary inflammation that could impair gas exchange.
This layered defence strategy allows the lungs to remain open, sterile, and functional despite constant exposure to the external environment.
Image: Section of the upper respiratory tract showing cilia, fine hair-like projections on the surface of epithelial cells that beat within a thin periciliary layer to move the overlying mucus. This coordinated motion helps clear trapped particles and pathogens from the airways as part of mucociliary clearance.
Beyond the Basics
The Mucociliary Escalator as a First-Line Defence
The mucociliary escalator forms the primary mechanical defence of the conducting airways. It relies on the coordinated action of cilia and a thin, well-hydrated mucus layer to trap inhaled particles and move them upward toward the pharynx for removal. Effective clearance depends on both the structural integrity of cilia and the physical properties of mucus.
Damage to ciliated epithelial cells, such as from cigarette smoke, air pollution, or viral infection, disrupts ciliary beating and slows mucus transport. Similarly, dehydration, chronic airway inflammation, and genetic disorders such as cystic fibrosis alter mucus composition, increasing its viscosity and reducing its mobility. When mucus becomes thick or stagnant, pathogens are retained within the airways, creating an environment that promotes bacterial growth, recurrent infection, and airway obstruction.
Alveolar Macrophages and Innate Immune Surveillance
Beyond the conducting airways, defence within the alveoli relies predominantly on alveolar macrophages. These specialised immune cells patrol the alveolar surface, phagocytosing inhaled particles, microorganisms, and cellular debris that escape proximal clearance mechanisms.
While alveolar macrophages are highly effective under normal conditions, their capacity is finite. In situations of overwhelming infection, inhalation injury, or extensive inflammation, the volume of debris may exceed their clearance ability. In such cases, macrophages release inflammatory mediators that recruit additional immune cells to the lung.
In severe inflammatory states, such as acute respiratory distress syndrome, macrophage activation contributes to increased capillary permeability. This allows protein-rich fluid to leak into the alveoli, disrupting the gas-exchange surface and compounding respiratory failure. Thus, a defence mechanism that is protective under normal conditions can become pathologically damaging when dysregulated.
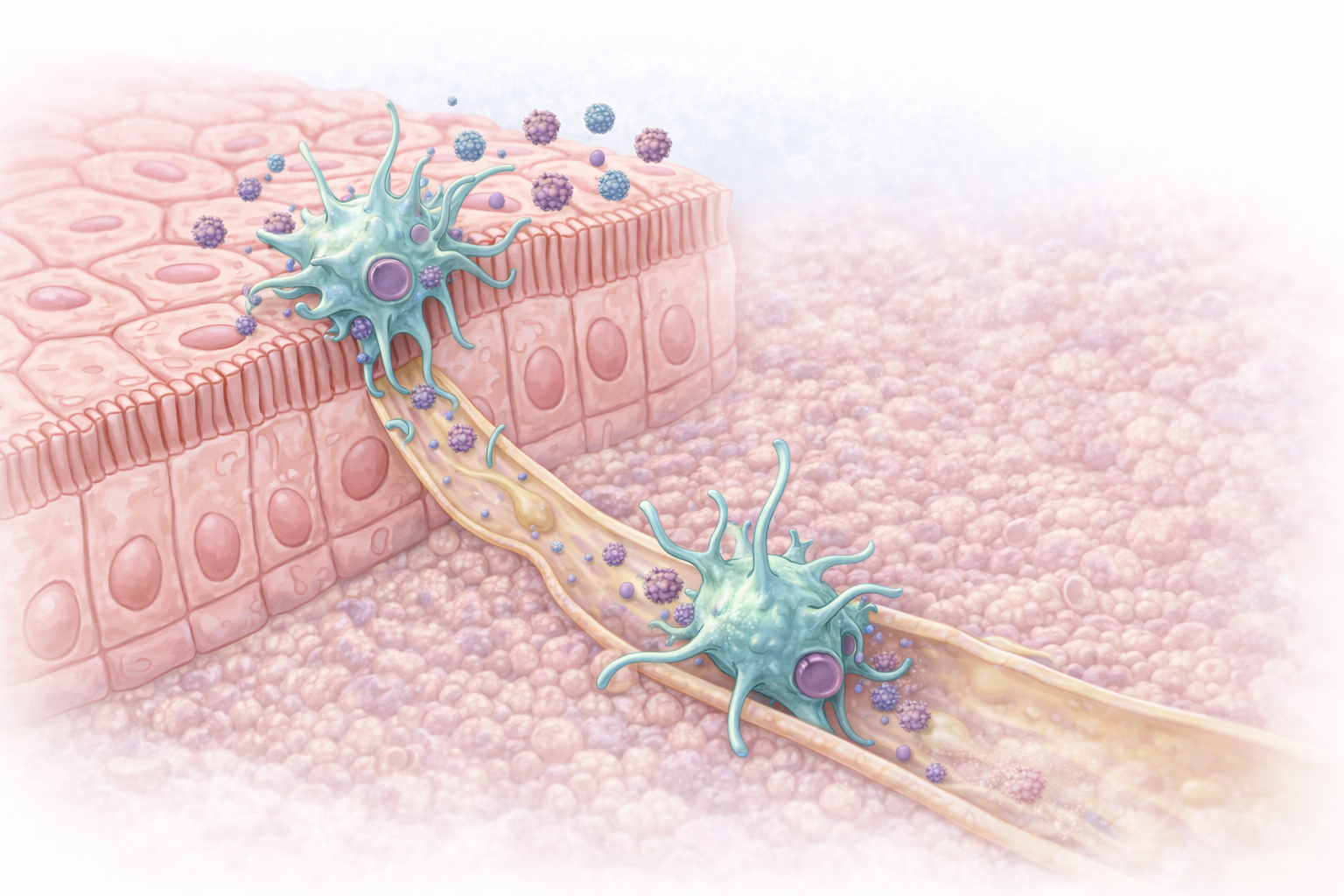
Image: Dendritic cells within the airway epithelium sample inhaled antigens and migrate through underlying tissue into lymphatic vessels, where they transport these antigens to lymphoid tissue to initiate antigen-specific immune responses.
Adaptive Immune Responses in the Respiratory Tract
The respiratory system also employs adaptive immune mechanisms to provide targeted and long-lasting protection against pathogens. Dendritic cells within the airway epithelium sample inhaled antigens and migrate to lymphoid tissue, where they present these antigens to T lymphocytes, initiating antigen-specific immune responses.
Secretory immunoglobulin A (IgA) plays a crucial role in mucosal immunity by binding pathogens and toxins, preventing their attachment to epithelial cells. This neutralisation occurs without provoking excessive inflammation, preserving airway integrity.
Cell-mediated immunity further supports respiratory defence. T lymphocytes identify and eliminate infected or damaged cells, limiting pathogen spread and promoting resolution of infection. However, dysregulation of adaptive immune responses can contribute to disease. In asthma, exaggerated immune responses to otherwise harmless antigens lead to chronic airway inflammation, bronchoconstriction, and mucus hypersecretion, illustrating how immune mechanisms can become maladaptive.
Integration and Balance in Respiratory Defence
Effective respiratory defence depends on the coordinated interaction between mechanical barriers, innate immunity, and adaptive immune responses. Each layer provides protection while minimising tissue damage and preserving gas exchange.
When these systems are compromised or excessively activated, susceptibility to infection and inflammatory lung disease increases. Understanding this balance is essential for interpreting respiratory pathology and appreciating how protective mechanisms can become drivers of disease.
Clinical Connections
When the respiratory defence mechanisms are disrupted, the respiratory system becomes vulnerable to infection and disease processes. Smoking damages multiple layers of airway protection at once:
it paralyses cilia
increases the viscosity and volume of mucus
suppresses alveolar macrophage activity
As a result, bacteria and viruses are not cleared efficiently and remain in contact with airway tissues, increasing the risk of bronchitis, pneumonia, and chronic obstructive pulmonary disease. Even short-term smoking during illness can worsen secretion retention and delay recovery.
Viral infections such as influenza and RSV injure the epithelial lining of the airways, stripping away the protective mucociliary layer and exposing deeper tissues. This explains why patients often deteriorate several days after a viral illness, developing secondary bacterial pneumonia once normal defences have been breached. In cystic fibrosis, abnormally thick, dehydrated mucus cannot be moved effectively by cilia, leading to airway plugging, chronic bacterial colonisation, and progressive lung damage driven by persistent inflammation.
Loss or failure of immune defence dramatically changes infection risk:
Immunocompromised patients (chemotherapy, transplant, HIV, long-term steroids) have reduced macrophage and lymphocyte activity
Opportunistic organisms such as Pneumocystis jirovecii, Aspergillus, and atypical mycobacteria can cause severe, rapidly progressive pneumonia
Fever, hypoxia, or subtle respiratory changes may be the only early signs of infection
In asthma, the immune system itself becomes part of the problem. Airway exposure to allergens or irritants triggers an exaggerated inflammatory response, leading to bronchoconstriction (airway narrowing), mucosal swelling, and mucus hypersecretion. These changes physically obstruct airflow and overwhelm normal clearance mechanisms, explaining why even minor viral infections can precipitate life-threatening asthma exacerbations.
Acute respiratory distress syndrome (ARDS) represents the catastrophic failure of pulmonary defence and repair systems. Widespread inflammation damages the alveolar–capillary membrane, disrupts surfactant, and causes alveolar collapse and flooding. Gas exchange becomes severely impaired, and even high levels of oxygen may not correct hypoxia. Recognising early signs of inflammatory lung injury, such as rising oxygen requirements, increased work of breathing, and new infiltrates on imaging, is critical for timely escalation of care.
Concept Check
How does the mucociliary escalator protect the lungs from infection?
Why do smokers have reduced airway defence?
How do alveolar macrophages contribute to both protection and pathology?
Why do viral infections increase the risk of secondary bacterial pneumonia?
How do immune responses contribute to the symptoms of asthma?