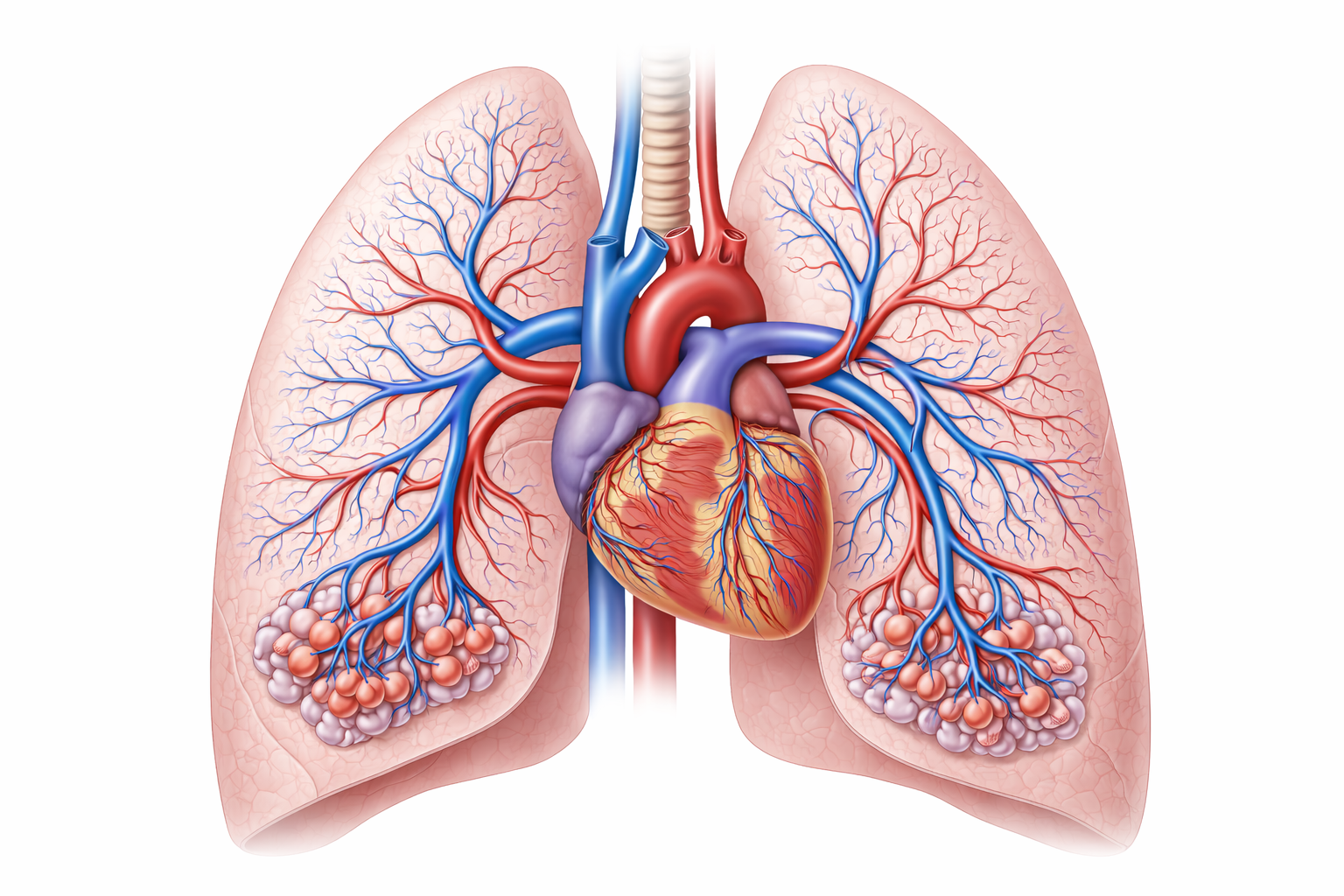
Pulmonary Circulation: Structure and Function
Pulmonary circulation is a unique vascular system designed specifically for gas exchange. Unlike systemic circulation, which delivers oxygenated blood at high pressure to the tissues, the pulmonary circuit transports deoxygenated blood at low pressure to the lungs, allowing efficient exchange of oxygen and carbon dioxide across the respiratory membrane. The delicate capillary networks that surround the alveoli must balance sufficient blood flow to maintain gas exchange while protecting the thin membrane from damage. Understanding pulmonary circulation provides insight into conditions such as pulmonary hypertension, pulmonary embolism, heart failure, and acute respiratory distress syndrome.
What You Need to Know
The pulmonary circulation is a low-pressure, high-flow system designed specifically for gas exchange rather than nutrient delivery. Deoxygenated blood leaves the right ventricle through the pulmonary trunk and enters the right and left pulmonary arteries, which branch repeatedly as they travel through the lungs. These vessels become progressively smaller until they form an extensive network of pulmonary capillaries that tightly surround the alveoli. The capillaries are so narrow that red blood cells pass through in single file, ensuring that each cell is exposed to alveolar air for efficient oxygen uptake and carbon dioxide removal. After gas exchange occurs, blood flows into pulmonary venules and veins, which return fully oxygenated blood to the left atrium for distribution to the rest of the body.
Unlike the systemic circulation, pulmonary vessels operate under much lower pressures. Mean pulmonary arterial pressure is only about one-sixth of systemic arterial pressure. This protects the thin alveolar–capillary membrane from damage and prevents fluid from being forced out of the blood and into the alveoli, which would interfere with oxygen diffusion. The low-pressure design also allows the lungs to remain compliant and flexible, a critical feature for effective ventilation.
Several unique features make pulmonary circulation highly adaptable:
Capillary recruitment - previously unperfused capillaries open during exercise, increasing surface area for gas exchange
Capillary recruitment - previously unperfused capillaries open during exercise, increasing surface area for gas exchange
Capillary distension - existing vessels stretch to carry more blood without large pressure rises
Minimal resistance - wide, compliant vessels allow large changes in flow with small pressure changes
These mechanisms mean that when cardiac output increases, such as during exercise or stress, pulmonary blood flow can increase several-fold without causing pulmonary hypertension. This allows oxygen uptake to keep pace with metabolic demand while maintaining the integrity of the respiratory membrane.
Image: The pulmonary circulation carries deoxygenated blood from the right side of the heart to the lungs, where it passes through an extensive capillary network surrounding the alveoli before returning oxygenated blood to the left side of the heart.
Beyond the Basics
Hypoxic Pulmonary Vasoconstriction and Flow Redistribution
A key feature of the pulmonary circulation is hypoxic pulmonary vasoconstriction (HPV). Unlike systemic vessels, which dilate in response to hypoxia, pulmonary arterioles constrict when alveolar oxygen tension falls. This local response reduces blood flow to poorly ventilated alveoli and redirects perfusion toward regions with higher oxygen availability.
Under normal conditions, HPV is a highly adaptive mechanism that optimises ventilation–perfusion matching and preserves arterial oxygenation. By selectively diverting blood away from hypoxic alveoli, the lungs maximise the efficiency of gas exchange without requiring changes in cardiac output or ventilation.
However, when hypoxia becomes widespread rather than regional, such as in acute respiratory distress syndrome, severe pneumonia, or exposure to high altitude, vasoconstriction occurs across large portions of the pulmonary vasculature. This global increase in pulmonary vascular resistance raises pulmonary artery pressures and substantially increases the workload placed on the right ventricle. Over time, this can lead to right ventricular strain or failure, highlighting the close functional coupling between the pulmonary circulation and the heart.
Pulmonary Capillary Pressures and Oedema Formation
Pulmonary capillaries are uniquely structured to support gas exchange, with thin walls and low baseline pressures. While this design facilitates diffusion, it also makes the pulmonary circulation particularly vulnerable to pressure-related injury.
When pulmonary capillary hydrostatic pressure rises, fluid is forced out of the capillaries into the interstitial space and, if severe, into the alveoli. This process produces pulmonary oedema, which disrupts both ventilation and diffusion. In left-sided heart failure, elevated left atrial pressure is transmitted backward into the pulmonary veins and capillaries, raising hydrostatic pressure and promoting fluid transudation.
Pulmonary oedema can also occur in the absence of left heart failure. In high-altitude pulmonary oedema, exaggerated hypoxic pulmonary vasoconstriction produces uneven elevations in capillary pressure, leading to capillary leak and alveolar flooding. In both scenarios, the structural vulnerability of pulmonary capillaries directly links vascular pressures to impaired gas exchange.
Pulmonary Embolism and Acute Vascular Obstruction
Pulmonary embolism represents an abrupt interruption of pulmonary blood flow due to obstruction of a pulmonary artery by thrombus, fat, or air. When this occurs, ventilation to the affected alveoli may remain intact, but perfusion is suddenly reduced or absent. This creates regions of high ventilation–perfusion (V/Q) mismatch, also described as physiological dead space.
The immediate consequence is impaired oxygen uptake, as ventilated alveoli are unable to transfer oxygen into the bloodstream. Dead space ventilation increases, forcing the respiratory system to work harder to maintain adequate gas exchange. In addition, acute increases in pulmonary vascular resistance place sudden strain on the right ventricle, which is poorly adapted to handle high afterload.
Clinically, these combined effects explain the characteristic features of pulmonary embolism, including tachycardia, dyspnoea, pleuritic chest pain, and hypoxaemia. In massive embolism, cardiovascular collapse may occur due to acute right ventricular failure.
Structure–Function Integration in the Pulmonary Arteries
The pulmonary arteries are structurally distinct from systemic arteries. They have thinner walls, lower smooth muscle tone under resting conditions, and operate at much lower pressures. These features reflect their primary role as a low-resistance, high-flow circuit designed to accommodate the entire cardiac output while supporting gas exchange.
Pathology arises when this low-pressure system is exposed to sustained or sudden increases in resistance. Whether through hypoxic vasoconstriction, vascular obstruction, or backward pressure transmission from the left heart, increases in pulmonary arterial pressure have immediate consequences for both respiratory function and cardiac performance.
Clinical Connections
Pulmonary hypertension develops when pressure within the pulmonary arteries increases chronically. This may result from chronic hypoxia, vascular remodelling, recurrent pulmonary emboli, or left heart failure. Over time, the right ventricle must generate higher pressure to overcome this resistance, eventually leading to right-sided heart failure, or cor pulmonale.
Key mechanisms to recognise:
Increased pulmonary vascular resistance → rising pulmonary artery pressure
Increased afterload on the right ventricle → progressive right ventricular strain
Elevated pulmonary pressures → fluid transudation into lung tissue
Heart failure highlights the interdependence of cardiac and pulmonary function. When the left ventricle fails to pump effectively, blood accumulates in the pulmonary circulation, increasing pressure and causing fluid to leak into the alveoli. This fluid disrupts diffusion and produces symptoms such as orthopnoea, exertional dyspnoea, and crackles on auscultation.
Diseases that destroy pulmonary vessels, such as emphysema, reduce the available capillary bed, increasing vascular resistance and raising pulmonary pressures. This contributes to the development of pulmonary hypertension in advanced COPD.
Concept Check
Why is pulmonary blood pressure lower than systemic blood pressure?
How does hypoxic pulmonary vasoconstriction help optimise gas exchange?
Why does pulmonary oedema occur in left-sided heart failure?
How does pulmonary embolism affect V/Q matching?
What long-term effects can chronic pulmonary hypertension have on the heart?