AORTIC DISSECTION: A Catastrophic Separation of the Aortic Wall Layers
Aortic dissection is a medical emergency in which a tear in the aortic intima allows blood to enter the medial layer, creating a false lumen within the aortic wall. As blood tracks between layers, it separates the vessel wall longitudinally, compromising blood flow to vital organs and risking rupture.
Aortic dissection is strongly associated with chronic hypertension and connective tissue disorders that weaken the aortic wall. Without prompt diagnosis and management, mortality increases rapidly, particularly in dissections involving the ascending aorta.
What You Need to Know
Aortic dissection is a life-threatening emergency caused by a tear in the intima, the innermost layer of the aortic wall. Under systemic arterial pressure, blood forces its way into the media, the middle muscular layer, creating a false lumen that separates the wall layers apart. This false channel can extend proximally toward the heart or distally along the descending aorta, sometimes involving major arterial branches. The result is not simply bleeding within the wall, but a dynamic and unstable disruption of aortic structure that can rapidly compromise organ perfusion.
Several underlying factors weaken the aortic media before the tear occurs. Chronic hypertension is the most common contributor, producing long-term shear stress that accelerates medial degeneration. Connective tissue disorders such as Marfan syndrome or Ehlers–Danlos syndrome reduce structural strength by altering collagen or fibrillin integrity. Aortic aneurysm, bicuspid aortic valve, inflammatory aortitis and prior cardiac surgery also increase vulnerability. When the intimal tear develops, high-pressure blood flow drives the dissection plane forward with each cardiac cycle.
The clinical consequences depend on the extent and location of the dissection. The process can:
Obstruct branch vessels, reducing blood flow to the brain, myocardium, kidneys, spinal cord or limbs.
Cause acute aortic regurgitation if the aortic valve is disrupted.
Rupture outward through the adventitia, leading to catastrophic internal haemorrhage.
Patients classically describe sudden, severe chest or back pain that is sharp or tearing in character, though presentations vary. Some may develop neurological deficits, myocardial ischaemia, syncope or signs of shock if major vessels are compromised. Without urgent recognition and intervention, mortality increases rapidly, particularly in dissections involving the ascending aorta.
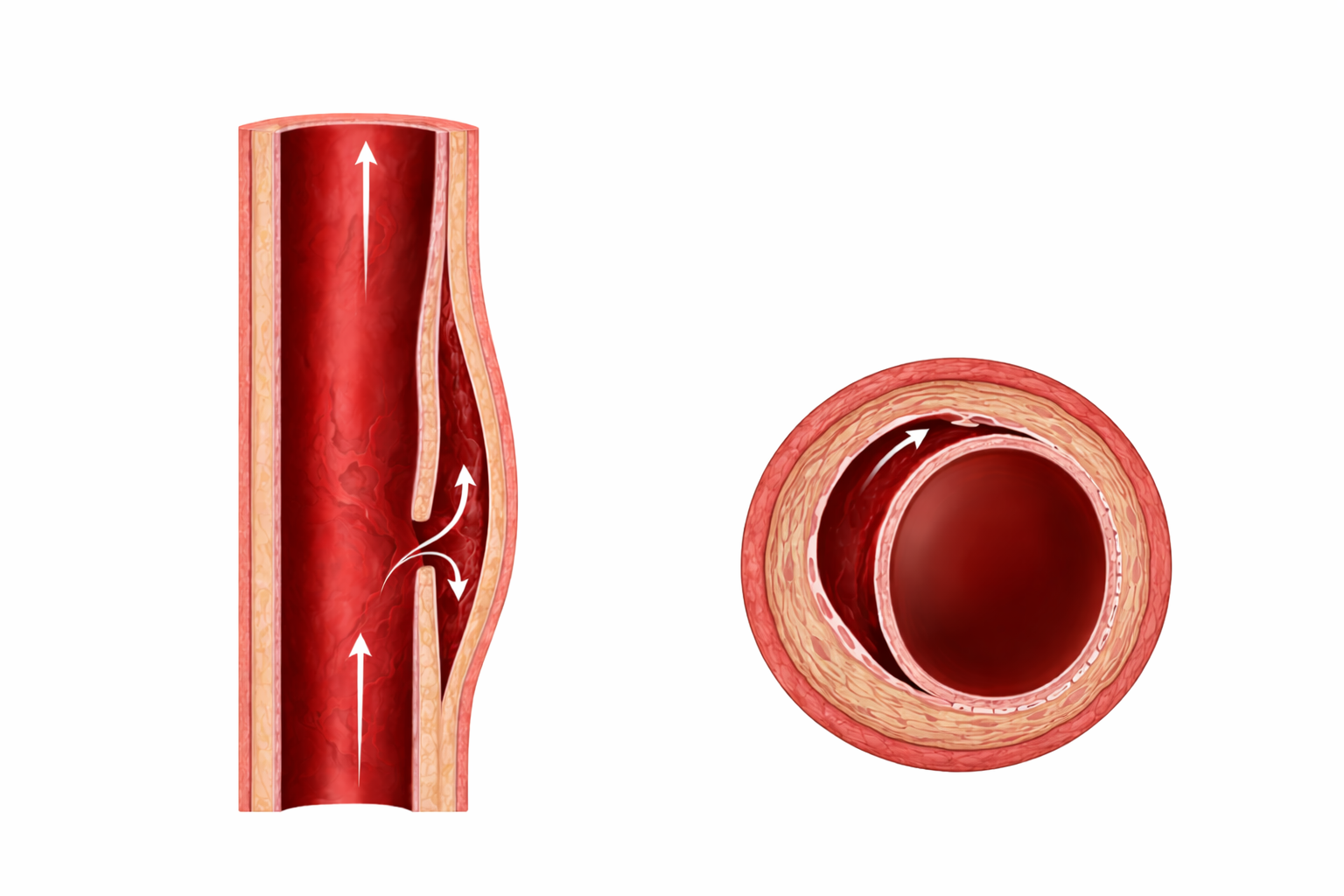
Image: An aortic dissection occurs when a tear in the inner layer of the vessel wall allows blood to enter and track between the layers, creating a false lumen. Blood flows through this newly formed channel alongside the true lumen, which can become compressed and reduce normal blood flow.
Beyond the Basics
Medial Degeneration and Structural Weakness
The structural vulnerability in aortic dissection begins within the media, the thick middle layer of the aortic wall composed of smooth muscle cells and elastic fibres arranged in lamellae. Chronic hypertension exposes the aorta to sustained shear stress, gradually fragmenting elastic fibres and reducing tensile strength. Over time, this leads to cystic medial degeneration, a process characterised by loss of smooth muscle cells and accumulation of mucoid extracellular material that weakens the wall’s structural cohesion.
Connective tissue disorders further compromise medial integrity. In Marfan syndrome, mutations affecting fibrillin impair elastic fibre formation, while in Ehlers–Danlos syndrome, abnormal collagen reduces tissue resilience. Even in individuals without inherited disorders, age-related degeneration and pre-existing aortic aneurysm increase susceptibility. The ascending aorta is particularly vulnerable because it experiences the highest pulsatile force during systole, amplifying mechanical stress at the point where the aorta exits the left ventricle.
Formation of the False Lumen
Once an intimal tear occurs, blood enters the medial layer under systemic arterial pressure and dissects longitudinally along the vessel wall. This creates a false lumen, a secondary channel within the aortic wall that may extend proximally toward the heart or distally along the descending aorta. The dissection flap separating the true and false lumens can shift dynamically with each cardiac cycle.
The haemodynamic consequences depend on how the false lumen interacts with the true lumen. Compression of the true lumen can reduce downstream perfusion, particularly during systole when pressure within the false lumen rises. In other cases, the dissection extends into branch vessels, producing either dynamic obstruction, where the flap intermittently blocks flow, or fixed obstruction, where the branch vessel origin is structurally narrowed. Occasionally, a distal re-entry tear allows blood to exit the false lumen, which may partially decompress it and reduce expansion. Without such decompression, progressive false lumen expansion increases wall tension and rupture risk.
Classification and Haemodynamic Impact
Aortic dissections are most commonly classified using the Stanford system, which categorises dissections based on ascending aortic involvement. Type A dissections involve the ascending aorta, regardless of distal extension, and represent a surgical emergency due to the high risk of life-threatening complications. Type B dissections are confined to the descending aorta and are often initially managed with aggressive blood pressure control unless complications such as malperfusion, rupture, or refractory pain develop.
In Type A dissections, involvement of the aortic root can distort the aortic valve annulus, leading to acute aortic regurgitation, where blood flows backward into the left ventricle during diastole. This sudden volume overload may precipitate acute pulmonary oedema or cardiogenic shock. Extension into the pericardial space can cause haemopericardium, accumulation of blood within the pericardial sac, resulting in cardiac tamponade, a condition in which rising pericardial pressure restricts ventricular filling and rapidly reduces cardiac output.
Malperfusion Syndromes
Malperfusion occurs when the dissection compromises blood flow to branch vessels arising from the aorta. Unlike embolic disease, where a clot travels and occludes a distal vessel, malperfusion in aortic dissection is mechanical, caused by flap displacement or true lumen compression. The pattern of organ injury reflects which arterial branches are involved.
Renal artery involvement may cause acute kidney injury with rising creatinine and reduced urine output. Carotid artery compromise can lead to focal neurological deficits or stroke. If the dissection affects intercostal or spinal arteries supplying the spinal cord, spinal cord ischaemia may occur, potentially resulting in paraplegia. Mesenteric artery involvement can produce bowel ischaemia, presenting with severe abdominal pain that may be disproportionate to early examination findings. These complications illustrate how a single intimal tear can trigger widespread systemic consequences through altered vascular flow rather than primary thrombotic occlusion.
Clinical Connections
Aortic dissection most often presents with sudden, severe pain that reaches maximal intensity within seconds. Patients frequently describe it as tearing, ripping, or sharp, and the location may shift as the dissection extends along the aorta. Anterior chest pain is more typical of ascending aortic involvement, whereas interscapular or back pain is more common in descending dissections. A difference in blood pressure between limbs, pulse deficits, new neurological findings, or unexplained hypotension should immediately raise concern for branch vessel compromise or rupture.
Aortic dissection can be classified based on location an extent of tear:
Ascending (Stanford Type A) involvement: chest pain, possible new diastolic murmur from acute aortic regurgitation, syncope, or haemodynamic instability due to tamponade.
Descending (Stanford Type B) involvement: back or abdominal pain, hypertension, and signs of limb or organ malperfusion.
Branch vessel compromise: focal neurological deficits, acute kidney injury, mesenteric ischaemia, or limb ischaemia caused by mechanical obstruction of arterial flow.
Diagnosis is confirmed with CT angiography, which visualises the intimal flap, differentiates true and false lumens, and identifies extension into branch vessels. Transoesophageal echocardiography may be used in unstable patients when rapid bedside imaging is required. Initial management focuses on immediate reduction of shear stress by controlling heart rate and blood pressure, typically with intravenous beta blockers to reduce the force of ventricular contraction, followed by vasodilators if additional blood pressure control is needed.
Type A dissections require urgent surgical repair because of the high risk of rupture, tamponade, and coronary involvement. Type B dissections are often managed medically with strict haemodynamic control unless complications such as persistent pain, malperfusion, aneurysmal expansion, or rupture develop. Ongoing monitoring is critical, particularly after surgical repair, as complications may include recurrent dissection, bleeding, organ dysfunction, or arrhythmias.
Concept Check
Why does chronic hypertension predispose patients to aortic dissection?
How does formation of a false lumen impair organ perfusion?
Why are ascending aortic dissections more dangerous than descending dissections?
How can aortic dissection lead to acute aortic regurgitation or tamponade?
Why is heart rate control as important as blood pressure control in acute management?