MITRAL STENOSIS: Obstruction of Left Ventricular Filling
Mitral stenosis (MS) is a narrowing of the mitral valve opening that impedes blood flow from the left atrium to the left ventricle during diastole. As the valve becomes thickened, calcified or fused, the left atrium must generate higher pressures to push blood through the restricted orifice. Over time, this leads to left atrial enlargement, elevated pulmonary pressures and progressive pulmonary congestion.
The most common cause of mitral stenosis worldwide is rheumatic heart disease, where autoimmune-mediated inflammation produces leaflet thickening, commissural fusion and chordal shortening. In developed countries, degenerative calcific disease is now increasingly recognised. Mitral stenosis often progresses gradually, with symptoms emerging only after the valve area becomes significantly reduced.
What You Need to Know
Mitral stenosis is a narrowing of the mitral valve that obstructs blood flow from the left atrium into the left ventricle during diastole. Under normal conditions, the mitral valve opens widely to allow passive ventricular filling when the ventricle relaxes. When the valve leaflets become thickened, stiff, or fused, as commonly occurs following rheumatic heart disease, blood flow is restricted and a diastolic pressure gradient develops across the valve. This means left atrial pressure must rise in order to push blood into the ventricle, setting off a cascade of haemodynamic changes.
As left atrial pressure increases, the atrium gradually dilates to accommodate the higher volume and pressure load. A dilated atrium is electrically unstable and prone to atrial fibrillation, which removes coordinated atrial contraction and further reduces ventricular filling. Because left ventricular preload falls, stroke volume and cardiac output become limited, particularly during exercise when diastolic filling time shortens. At the same time, elevated left atrial pressure is transmitted backward into the pulmonary veins and capillaries, leading to pulmonary congestion and progressive breathlessness.
The core pathophysiological consequences can be summarised as follows:
Obstructed diastolic flow increases left atrial pressure and creates a pressure gradient across the mitral valve.
Left atrial dilation predisposes to atrial fibrillation and thrombus formation.
Pulmonary venous congestion causes dyspnoea and, over time, pulmonary hypertension with right ventricular strain.
With chronic elevation of pulmonary pressures, the right ventricle must work harder to overcome increased resistance in the pulmonary circulation. This can lead to right ventricular hypertrophy and eventually right-sided heart failure. Although the primary problem is valvular obstruction, the long-term effects involve both sides of the heart and the pulmonary vasculature.
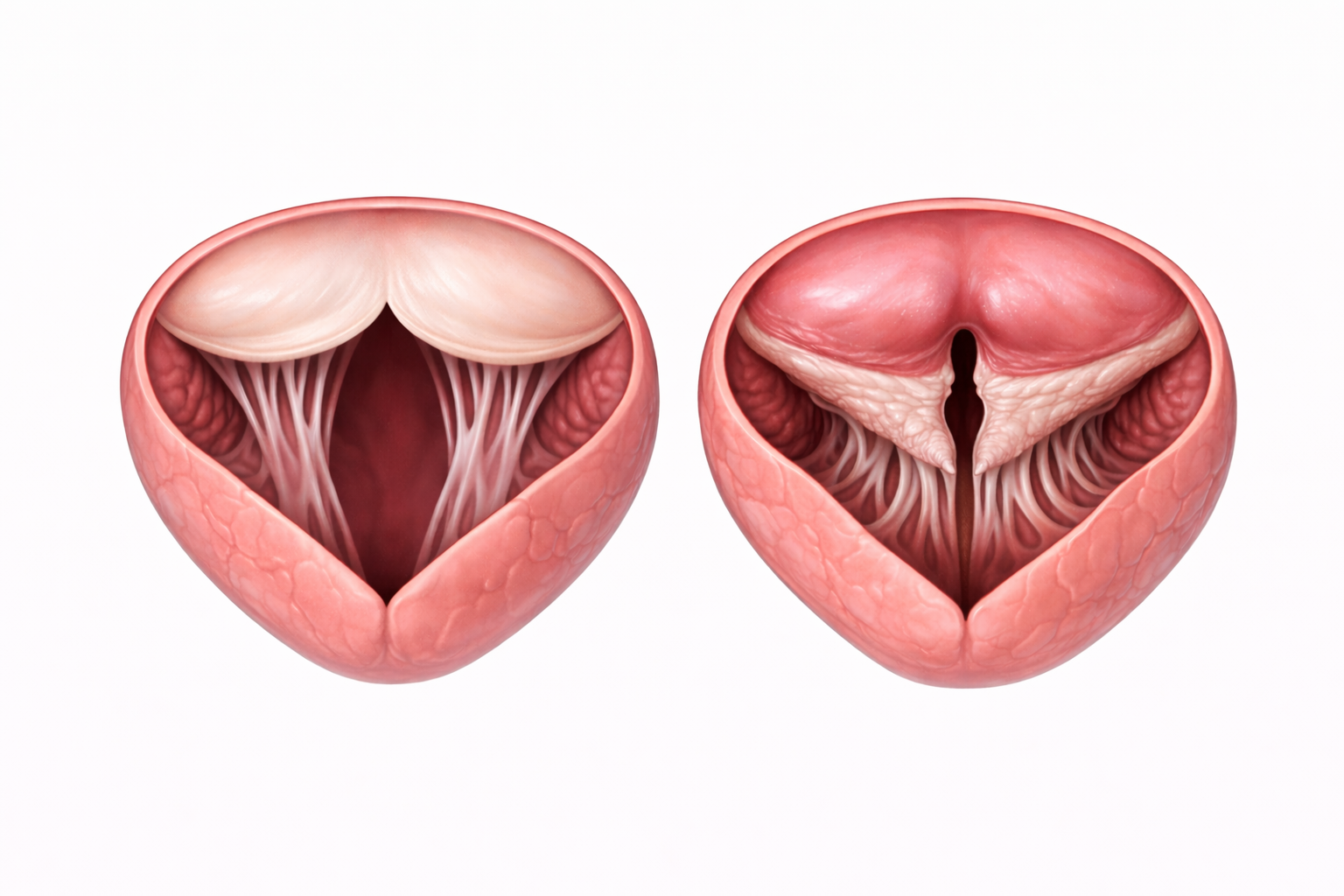
Image: The valve on the right is stenosed, with thickened, fused leaflets creating a narrowed opening that restricts blood flow compared to the normal valve on the left.
Beyond the Basics
Rheumatic Disease and Structural Valve Changes
Mitral stenosis most commonly develops as a late consequence of rheumatic fever, an immune-mediated inflammatory response to group A streptococcal infection. During the acute phase, inflammation affects the mitral valve leaflets and the subvalvular apparatus, including the chordae tendineae and papillary muscles. Repeated or unresolved inflammation promotes progressive fibrosis, commissural fusion, and leaflet thickening. Over time, the normally thin, pliable valve becomes rigid and narrowed, often assuming a funnel-shaped configuration with a reduced orifice area.
The subvalvular structures are also affected. Chordae tendineae may shorten, fuse, and lose flexibility, tethering the leaflets and further limiting their ability to open during diastole. In advanced disease, dystrophic calcification develops within the valve tissue, increasing stiffness and reducing compliance. The result is a fixed mechanical obstruction that does not adapt to changes in flow demand. Even at rest, blood flow across the valve is restricted, and during periods of increased cardiac output, the obstruction becomes more haemodynamically significant.
Haemodynamic Consequences and Pulmonary Congestion
The narrowed valve creates resistance to diastolic filling, requiring higher left atrial pressures to maintain forward flow into the ventricle. This persistent elevation in left atrial pressure is transmitted backward into the pulmonary veins and capillary bed, increasing hydrostatic pressure within the pulmonary circulation. Fluid shifts into the interstitial and alveolar spaces, reducing lung compliance and impairing gas exchange. Clinically, this manifests as exertional dyspnoea, orthopnoea, and paroxysmal nocturnal dyspnoea as pulmonary congestion progresses.
Chronic elevation of pulmonary venous pressure can trigger reactive pulmonary arterial vasoconstriction, followed by structural remodelling of the pulmonary vasculature, including medial hypertrophy and intimal thickening. This transition from passive congestion to established pulmonary hypertension increases right ventricular afterload. As right ventricular pressure overload develops, right ventricular hypertrophy and eventual dilation may occur, progressing to right-sided heart failure in advanced disease.
Reduced Cardiac Output and Exercise Intolerance
Because ventricular filling is restricted, stroke volume remains limited. Cardiac output may be relatively preserved at rest in mild disease, but the inability to increase preload becomes evident during exertion. Tachycardia shortens diastole, further reducing the time available for blood to cross the stenotic valve. The combination of reduced filling time and fixed obstruction leads to a disproportionate fall in cardiac output during activity, contributing to fatigue, reduced exercise tolerance, and exertional light-headedness.
Atrial fibrillation has a particularly profound haemodynamic impact in mitral stenosis. The coordinated atrial contraction that normally contributes to late diastolic filling, often referred to as the atrial kick, becomes absent. In the presence of a narrowed valve, this loss of atrial contribution can significantly reduce ventricular filling and precipitate acute decompensation. Rapid ventricular rates further shorten diastole, compounding the reduction in cardiac output.
Left Atrial Enlargement and Thromboembolic Risk
Sustained pressure overload causes progressive enlargement of the left atrium. As the chamber dilates, blood flow becomes increasingly sluggish, particularly within the left atrial appendage. Stasis of blood promotes thrombus formation through local activation of coagulation pathways. The presence of atrial fibrillation further increases thromboembolic risk by eliminating organised atrial contraction and promoting turbulent flow.
Thrombi formed within the left atrium may embolise into the systemic circulation, with cerebral vessels being a common destination, resulting in ischaemic stroke. Other systemic embolic events, including limb or visceral ischaemia, may also occur. This risk profile explains why anticoagulation is indicated in patients with mitral stenosis who develop atrial fibrillation or demonstrate significant atrial enlargement, even in the absence of prior embolic events.
Clinical Connections
Mitral stenosis typically presents with symptoms that are consistent with rising left atrial pressure and pulmonary venous congestion. Exertional dyspnoea is often the earliest complaint, progressing to orthopnoea and paroxysmal nocturnal dyspnoea as pulmonary pressures increase. Fatigue and reduced exercise tolerance result from limited cardiac output, particularly during activity when diastolic filling time shortens. On auscultation, a low-pitched diastolic murmur best heard at the apex, often preceded by an opening snap, is characteristic and corresponds to turbulent flow across the narrowed valve. In some cases, haemoptysis occurs due to rupture of bronchial veins exposed to chronically elevated pulmonary venous pressures.
Certain clinical patterns help differentiate stable from progressive disease:
New or worsening breathlessness may indicate rising pulmonary pressures or the onset of atrial fibrillation.
Rapid, irregular pulse suggests atrial fibrillation, which can abruptly worsen symptoms due to loss of coordinated atrial contraction.
Peripheral oedema, hepatomegaly or ascites signal progression to pulmonary hypertension with secondary right-sided heart strain.
Echocardiography confirms the diagnosis by directly visualising leaflet thickening, commissural fusion, restricted mobility and calcification. It also measures mitral valve area and the transvalvular gradient, allowing grading of severity and assessment of suitability for intervention. Management targets both haemodynamic consequences and complications. Diuretics reduce pulmonary congestion, rate control improves diastolic filling in atrial fibrillation, and anticoagulation reduces thromboembolic risk in patients with atrial fibrillation or significant atrial enlargement. In selected patients with favourable valve anatomy, percutaneous balloon mitral valvotomy can relieve obstruction by separating fused commissures, while advanced or heavily calcified disease may require surgical repair or valve replacement. Ongoing monitoring is essential to detect progression, development of pulmonary hypertension, new arrhythmias, or signs of systemic embolisation.
Concept Check
How does narrowing of the mitral valve impair left ventricular filling?
Why does mitral stenosis lead to elevated pulmonary venous pressures?
What structural changes occur in rheumatic mitral stenosis?
Why is atrial fibrillation particularly dangerous in patients with mitral stenosis?
How does pulmonary hypertension develop as a complication of MS?