TRICUSPID REGURGITATION
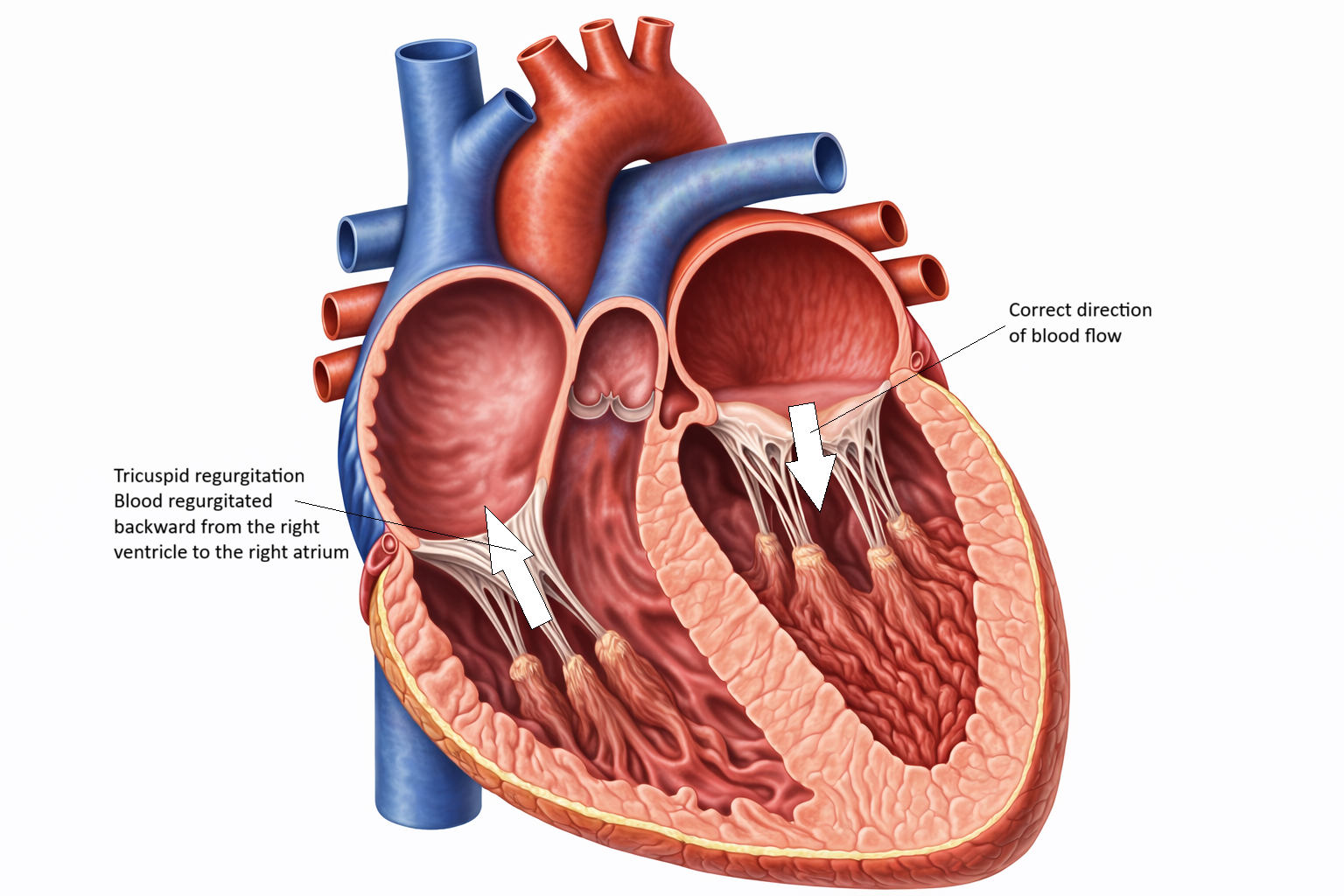
Tricuspid regurgitation (TR) occurs when the tricuspid valve fails to close completely during right ventricular systole, allowing blood to flow backward into the right atrium. While often overshadowed by left-sided valve diseases, tricuspid regurgitation significantly affects venous return, systemic congestion and right-sided cardiac function.
TR is most commonly functional (secondary), meaning the valve leaflets are structurally normal but the right ventricle or tricuspid annulus is dilated, preventing the leaflets from coapting effectively. Less commonly, primary tricuspid regurgitation arises from intrinsic pathology of the valve leaflets, such as infective endocarditis, rheumatic disease or carcinoid syndrome. Regardless of cause, the consequence is chronic volume overload of the right atrium and right ventricle.
What You Need to Know
Tricuspid regurgitation is the backflow of blood from the right ventricle into the right atrium during systole due to incompetence of the tricuspid valve. Under normal conditions, the valve closes firmly when the right ventricle contracts, ensuring that blood is propelled forward into the pulmonary artery. When the valve fails to coapt properly, part of the right ventricular stroke volume is ejected backward. This creates a volume overload state within the right atrium, which gradually enlarges to accommodate the regurgitant flow.
As regurgitation persists, the right ventricle is also exposed to chronic volume overload because it must handle both the normal venous return and the blood that refluxes back into the atrium and returns again during the next cardiac cycle. Over time, the right ventricle dilates and may lose contractile efficiency. Although tricuspid regurgitation can initially be well tolerated, progressive chamber dilation reduces effective forward flow into the pulmonary circulation and limits oxygen delivery to the lungs.
The key haemodynamic consequences include:
Right atrial enlargement due to regurgitant volume during systole.
Right ventricular dilation from chronic volume overload.
Rising systemic venous pressure as forward flow declines.
Elevated systemic venous pressure leads to congestion in organs drained by the systemic venous system. Patients commonly develop peripheral oedema, abdominal distension from ascites, hepatic congestion causing right upper quadrant discomfort, and visible jugular venous distension. Fatigue occurs as effective pulmonary blood flow and overall cardiac output decline. Although the primary abnormality lies in the right heart, the systemic effects are often the most clinically apparent features.
Image: Tricuspid regurgitation: the tricuspid valve does not close fully during ventricular contraction, allowing blood to flow backward from the right ventricle into the right atrium.
Beyond the Basics
Secondary (Functional) Tricuspid Regurgitation
Most tricuspid regurgitation is functional rather than due to primary leaflet damage. It develops as a consequence of right ventricular dilation or chronically elevated pulmonary pressures. Pulmonary hypertension, left-sided heart failure, and chronic lung disease increase right ventricular afterload, meaning the right ventricle must generate higher pressure to eject blood into the pulmonary circulation. Over time, this pressure load causes right ventricular remodelling and dilation.
As the ventricle enlarges, the tricuspid annulus, the fibrous ring that anchors the valve leaflets, stretches. This annular dilation prevents the leaflets from meeting centrally during systole, disrupting coaptation despite structurally normal valve tissue. Regurgitation then develops as a mechanical consequence of chamber geometry rather than intrinsic valve disease. Functional tricuspid regurgitation illustrates how left-sided pathology or pulmonary vascular disease can progressively destabilise right heart function, eventually contributing to right-sided heart failure.
Primary Tricuspid Regurgitation
Primary tricuspid regurgitation results from direct structural damage to the valve leaflets or subvalvular apparatus. Infective endocarditis can destroy or perforate leaflets, particularly in individuals with intravenous drug use where the tricuspid valve is exposed to bloodstream pathogens. Rheumatic disease may cause leaflet thickening and retraction. Carcinoid syndrome, a condition associated with serotonin-secreting tumours, leads to fibrous plaque deposition on the valve, restricting mobility and preventing adequate closure.
Other causes include trauma, congenital malformations such as Ebstein anomaly, and iatrogenic injury from intracardiac pacing or defibrillator leads that interfere with leaflet motion. In these cases, regurgitation arises independently of annular dilation, although progressive right atrial and ventricular enlargement commonly develops over time as a secondary adaptation to chronic volume overload.
Volume Overload and Chamber Remodelling
During each systolic contraction, a proportion of right ventricular stroke volume regurgitates into the right atrium. The right atrium adapts by enlarging, often accommodating substantial regurgitant volumes without dramatic increases in pressure initially. This compensatory dilation helps maintain venous return in early disease but creates electrical instability within the atrial myocardium, increasing susceptibility to atrial fibrillation.
The right ventricle also undergoes progressive dilation because it must handle both systemic venous return and the recirculated regurgitant volume. Early dilation may preserve stroke volume through the Frank–Starling mechanism, where increased fibre stretch enhances contractile force. However, chronic stretch eventually impairs myocardial efficiency and contractility. As right ventricular function declines, forward pulmonary blood flow falls, reducing left ventricular preload and contributing to a global reduction in cardiac output.
Systemic Venous Congestion
Tricuspid regurgitation primarily manifests through systemic venous congestion rather than pulmonary oedema, which is more typical of left-sided valve disease. Elevated right atrial pressure is transmitted backward into the vena cavae and systemic venous circulation. This results in jugular venous distension, hepatomegaly, ascites, and peripheral oedema. The liver is particularly vulnerable to chronic congestion. Sustained hepatic venous hypertension leads to sinusoidal dilation and hepatocyte injury, a condition referred to as congestive hepatopathy. In advanced stages, fibrosis may develop, sometimes termed cardiac cirrhosis.
Venous congestion also affects the gastrointestinal tract. Oedema of the bowel wall can cause early satiety, anorexia, abdominal discomfort, and impaired nutrient absorption. These systemic effects often dominate the clinical picture and may progress insidiously over time.
Haemodynamic Consequences and Reduced Forward Flow
Because a portion of the right ventricular stroke volume is lost backward into the atrium, effective pulmonary blood flow declines. Reduced pulmonary circulation limits oxygen uptake and decreases left ventricular filling. Patients commonly experience fatigue, exercise intolerance, and exertional dyspnoea as cardiac output becomes insufficient to meet metabolic demand.
When pulmonary hypertension is the underlying driver, the combination of pressure overload and volume overload places substantial stress on the right ventricle. This dual burden accelerates right ventricular failure and worsens regurgitation in a self-perpetuating cycle. Over time, declining right ventricular contractility and persistent venous congestion mark the transition from compensated regurgitation to overt right-sided heart failure.
Clinical Connections
Tricuspid regurgitation often progresses insidiously, with symptoms emerging as systemic venous congestion becomes more pronounced. Patients may report increasing abdominal girth from ascites, bilateral ankle oedema, early satiety due to gut congestion, and right upper quadrant discomfort related to hepatic venous congestion. Fatigue can be profound, reflecting reduced forward pulmonary blood flow and diminished overall cardiac output. On examination, a holosystolic murmur heard best at the left lower sternal border may be detected, and its intensity can increase with inspiration due to increased venous return to the right heart.
Certain clinical patterns help identify progression and underlying cause:
Rising jugular venous pressure and peripheral oedema indicate worsening systemic venous congestion.
Hepatomegaly with tenderness suggests congestive hepatopathy from sustained right atrial pressure elevation.
New or worsening dyspnoea may reflect coexisting pulmonary hypertension or left-sided cardiac pathology driving functional regurgitation.
Echocardiography confirms the diagnosis by evaluating leaflet morphology, annular dilation, regurgitant jet size, and right ventricular size and systolic performance. It also helps distinguish primary leaflet pathology from functional regurgitation due to chamber enlargement. Management focuses first on addressing the underlying driver, such as optimising treatment of pulmonary hypertension or left-sided heart failure in functional tricuspid regurgitation. Diuretics are used to reduce venous congestion and improve symptoms, though they do not correct the structural problem. In more advanced or refractory cases, surgical repair, annuloplasty to reduce annular size, or transcatheter tricuspid interventions may be considered. Close clinical surveillance is important to detect progression toward overt right ventricular failure, escalating hepatic dysfunction, or recurrent fluid overload.
Concept Check
How does annular dilation contribute to secondary tricuspid regurgitation?
Why does chronic TR cause enlargement of both the right atrium and right ventricle?
How does tricuspid regurgitation create systemic venous congestion?
Why is atrial fibrillation common in long-standing TR?
How can left-sided heart failure contribute to the development of functional TR?