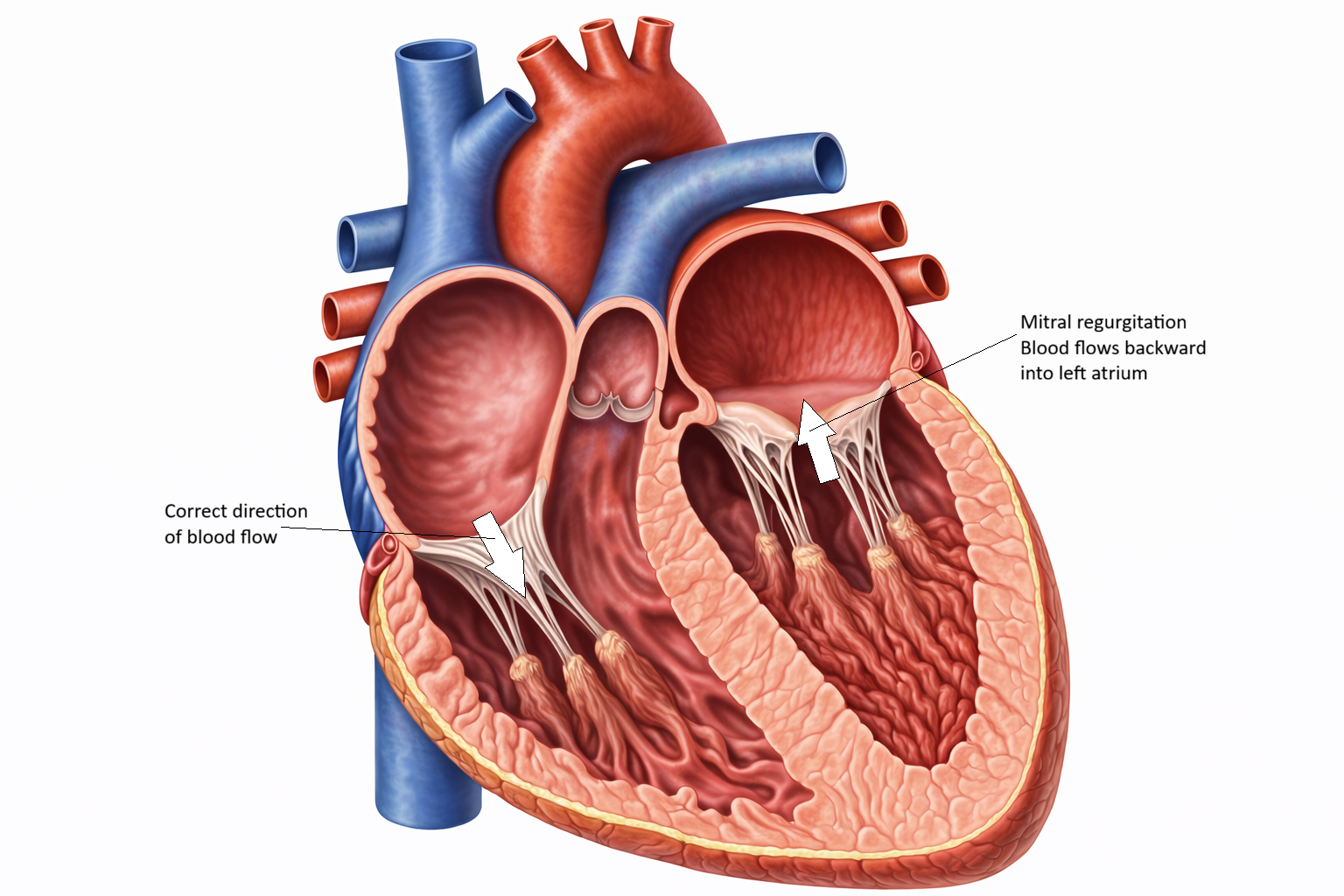
MITRAL REGURGITATION: Backward Flow of Blood Into the Left Atrium
Mitral regurgitation (MR) occurs when the mitral valve fails to close completely during systole, allowing a portion of the left ventricular stroke volume to leak backward into the left atrium. Unlike mitral stenosis, which restricts forward flow, MR creates a volume overload on both the left atrium and left ventricle.
Mitral regurgitation may be primary (degenerative), caused by structural abnormalities of the valve leaflets or chordae, or secondary (functional), caused by left ventricular dilation or dysfunction that distorts the valve apparatus. The clinical picture varies significantly depending on whether regurgitation is acute or chronic. Chronic MR develops slowly, allowing the atrium and ventricle time to remodel, whereas acute MR results in abrupt hemodynamic collapse and pulmonary oedema.
What You Need to Know
Mitral regurgitation is the backward flow of blood from the left ventricle into the left atrium during systole due to failure of the mitral valve to close effectively. Under normal conditions, the mitral leaflets coapt firmly when the ventricle contracts, ensuring that all stroke volume is directed forward into the aorta. When leaflet prolapse, chordae tendineae rupture, papillary muscle dysfunction, or ventricular dilation disrupts this seal, blood is ejected in two directions simultaneously. This creates a volume overload state affecting both the left atrium and left ventricle.
In chronic mitral regurgitation, the left atrium gradually dilates to accommodate the increased regurgitant volume while limiting the rise in atrial pressure. The left ventricle also enlarges and develops eccentric hypertrophy, meaning the chamber dilates with proportional wall thickening, in order to maintain forward stroke volume. For a period of time, these structural adaptations preserve cardiac output despite ongoing regurgitation. However, persistent volume overload eventually leads to progressive ventricular dilation, reduced contractile efficiency, and declining systolic function.
The key haemodynamic patterns include:
Volume overload of the left atrium and ventricle due to regurgitant flow during systole.
Eccentric left ventricular hypertrophy as an adaptive response to chronic volume load.
Pulmonary congestion when left atrial pressure rises beyond compensatory limits.
Acute mitral regurgitation behaves very differently. Because the left atrium has not had time to dilate, regurgitant flow causes a rapid and marked rise in left atrial pressure. This sudden pressure increase is transmitted directly into the pulmonary circulation, producing acute pulmonary oedema and severe breathlessness. At the same time, effective forward stroke volume falls abruptly, which may lead to hypotension and cardiogenic shock.
Image: Mitral regurgitation occurs due to incomplete closure of the mitral valve allows blood to flow backward from the left ventricle into the left atrium during systole, reducing forward cardiac output and increasing atrial volume.
Beyond the Basics
Structural and Functional Causes of Mitral Regurgitation
Mitral regurgitation is broadly divided into primary and secondary forms based on whether the problem originates within the valve apparatus itself or as a consequence of ventricular remodelling. Primary mitral regurgitation results from intrinsic structural abnormalities of the leaflets, chordae tendineae, papillary muscles, or annulus. Mitral valve prolapse is a common cause and is characterised by redundant, thickened, or myxomatous leaflets that billow into the left atrium during systole. Elongation or rupture of the chordae tendineae can abruptly increase regurgitation severity. Rheumatic disease may cause leaflet thickening and distortion, while infective endocarditis can destroy leaflet tissue or supporting structures, producing acute or subacute regurgitation.
Secondary mitral regurgitation arises when the valve leaflets are structurally normal but fail to coapt because of left ventricular dilation or geometric distortion. As the ventricle enlarges, the mitral annulus stretches and the papillary muscles are displaced laterally and apically. This tethering effect prevents the leaflets from meeting centrally during systole. Ischaemic heart disease is a major contributor, particularly when papillary muscle dysfunction or infarction impairs coordinated leaflet support. In this setting, regurgitation reflects ventricular pathology rather than primary valvular damage.
Volume Overload and Chamber Remodelling
Chronic mitral regurgitation imposes a sustained volume load on both the left atrium and left ventricle. Because part of the stroke volume is lost backward into the atrium, the ventricle compensates by increasing total stroke volume to preserve forward cardiac output. This adaptive response leads to ventricular dilation and eccentric hypertrophy, in which chamber enlargement occurs alongside proportional wall thickening. The goal of this remodelling is to maintain wall stress and preserve systolic function despite ongoing regurgitation.
The left atrium enlarges to accommodate regurgitant volume while limiting the rise in atrial pressure. For many years, this adaptation can prevent significant pulmonary congestion. However, atrial dilation promotes electrical instability and increases the likelihood of atrial fibrillation. Loss of coordinated atrial contraction reduces ventricular filling efficiency and may precipitate symptom progression. Eventually, chronic volume overload leads to declining ventricular contractility, reduced forward output, and the transition to overt heart failure.
Acute Versus Chronic Mitral Regurgitation
Acute mitral regurgitation differs dramatically in haemodynamic behaviour from chronic disease. When regurgitation develops suddenly, such as after papillary muscle rupture following myocardial infarction or acute chordal rupture, the left atrium has not undergone compensatory dilation. The abrupt regurgitant volume causes a rapid and marked rise in left atrial pressure. This pressure is transmitted immediately into the pulmonary circulation, leading to acute pulmonary oedema and severe respiratory distress. Because effective forward stroke volume falls sharply, hypotension and cardiogenic shock may develop quickly.
Chronic mitral regurgitation progresses more gradually. Compensatory atrial and ventricular dilation delay the onset of symptoms, and individuals may remain asymptomatic despite significant structural enlargement. Clinical deterioration typically occurs when ventricular systolic function begins to decline or when atrial fibrillation develops, reducing filling efficiency and destabilising previously compensated haemodynamics.
Haemodynamic Consequences
Mitral regurgitation reduces the efficiency of left ventricular contraction because a portion of each systolic ejection does not contribute to systemic perfusion. During diastole, the enlarged ventricle receives both normal pulmonary venous return and the regurgitant volume that has re-entered the atrium, perpetuating the cycle of volume overload. Over time, rising left atrial pressure is transmitted backward into the pulmonary veins and capillaries, producing exertional dyspnoea and pulmonary congestion.
Persistent pulmonary venous hypertension may eventually trigger secondary pulmonary arterial changes, increasing right ventricular afterload. Meanwhile, declining forward ejection efficiency contributes to fatigue, reduced exercise tolerance, and other features of heart failure. The interplay between adaptive remodelling and eventual decompensation defines the clinical trajectory of chronic mitral regurgitation.
Clinical Connections
Mitral regurgitation often progresses silently for years, particularly in chronic disease where atrial and ventricular dilation initially compensate for regurgitant volume. Patients may report subtle symptoms such as palpitations from atrial enlargement, exertional dyspnoea due to rising left atrial pressure, or reduced exercise tolerance as forward cardiac output becomes less efficient. On examination, a pansystolic murmur best heard at the apex and radiating to the axilla is characteristic, reflecting continuous backflow from the left ventricle into the left atrium throughout systole. As ventricular dilation advances and systolic function begins to decline, symptoms of heart failure become more apparent.
Certain clinical changes signal progression from compensation to decompensation:
Increasing breathlessness or reduced exercise capacity may indicate rising pulmonary pressures or declining ventricular function.
New onset atrial fibrillation can abruptly worsen symptoms by reducing filling efficiency and increasing heart rate.
Signs of left ventricular dysfunction, such as fatigue and reduced peripheral perfusion, suggest failing compensatory mechanisms.
Echocardiography confirms the diagnosis and quantifies regurgitant severity by assessing jet size, regurgitant volume, effective regurgitant orifice area, chamber dimensions, and left ventricular systolic function. Serial imaging is important in chronic mitral regurgitation because intervention is ideally timed before irreversible ventricular dysfunction develops. Management includes afterload reduction to decrease regurgitant fraction, rate or rhythm control for atrial fibrillation, and diuretics to relieve pulmonary congestion. In many cases, definitive treatment involves surgical repair, which is preferred when feasible, or valve replacement. Close clinical monitoring is essential to detect early ventricular decline, escalating symptoms, or arrhythmias that alter haemodynamic stability and influence timing of intervention.
Concept Check
Why does mitral regurgitation create a volume overload rather than a pressure overload?
How does the left ventricle adapt during chronic MR, and why do these adaptations eventually fail?
What are the key differences between primary and secondary MR?
Why is acute mitral regurgitation more dangerous than chronic MR?
How does left atrial dilation contribute to clinical complications in MR?