CARDIAC ARRHYTHMIAS: An Overview
Cardiac arrhythmias are abnormalities in the rate, rhythm or conduction of electrical impulses through the heart. They range from benign variations to life-threatening disturbances that impair cardiac output or predispose to sudden cardiac death. Arrhythmias arise when electrical impulses originate from abnormal foci, travel through abnormal conduction pathways or encounter tissue that cannot conduct properly. The consequences depend on the heart rate, the efficiency of mechanical contraction and the underlying cardiac condition.
What You Need to Know
Cardiac arrhythmias are disturbances in the heart’s normal rhythm caused by abnormalities in impulse generation, impulse conduction, or both. Under normal conditions, the sinoatrial (SA) node initiates electrical impulses that spread across the atria, pass through the atrioventricular (AV) node, and travel down the His–Purkinje system to coordinate ventricular contraction. This sequence ensures efficient atrial filling of the ventricles followed by organised ventricular ejection. When this electrical pathway is disrupted, the result may be a rhythm that is too fast, too slow, irregular, or chaotic.
Arrhythmias arise from a range of underlying mechanisms. Structural heart disease can create areas of fibrosis that disrupt conduction. Myocardial ischaemia alters cellular ion balance and membrane stability. Electrolyte disturbances, particularly involving potassium, magnesium, or calcium, interfere with normal depolarisation and repolarisation. Autonomic imbalance, medications, toxins, and congenital conduction abnormalities may also trigger or sustain abnormal rhythms. The clinical impact depends on both the type of arrhythmia and the haemodynamic stability of the individual.
The major consequences of arrhythmias can be grouped into three broad patterns:
Reduced cardiac output due to very fast, very slow, or uncoordinated ventricular contraction.
Thromboembolic risk, especially in atrial fibrillation where ineffective atrial contraction promotes blood stasis.
Sudden cardiac death risk, particularly with sustained ventricular tachycardia or ventricular fibrillation.
Some arrhythmias cause only mild palpitations or remain asymptomatic, while others rapidly compromise perfusion and require urgent intervention. Understanding the underlying mechanism helps predict clinical risk, guide monitoring, and inform appropriate management strategies.
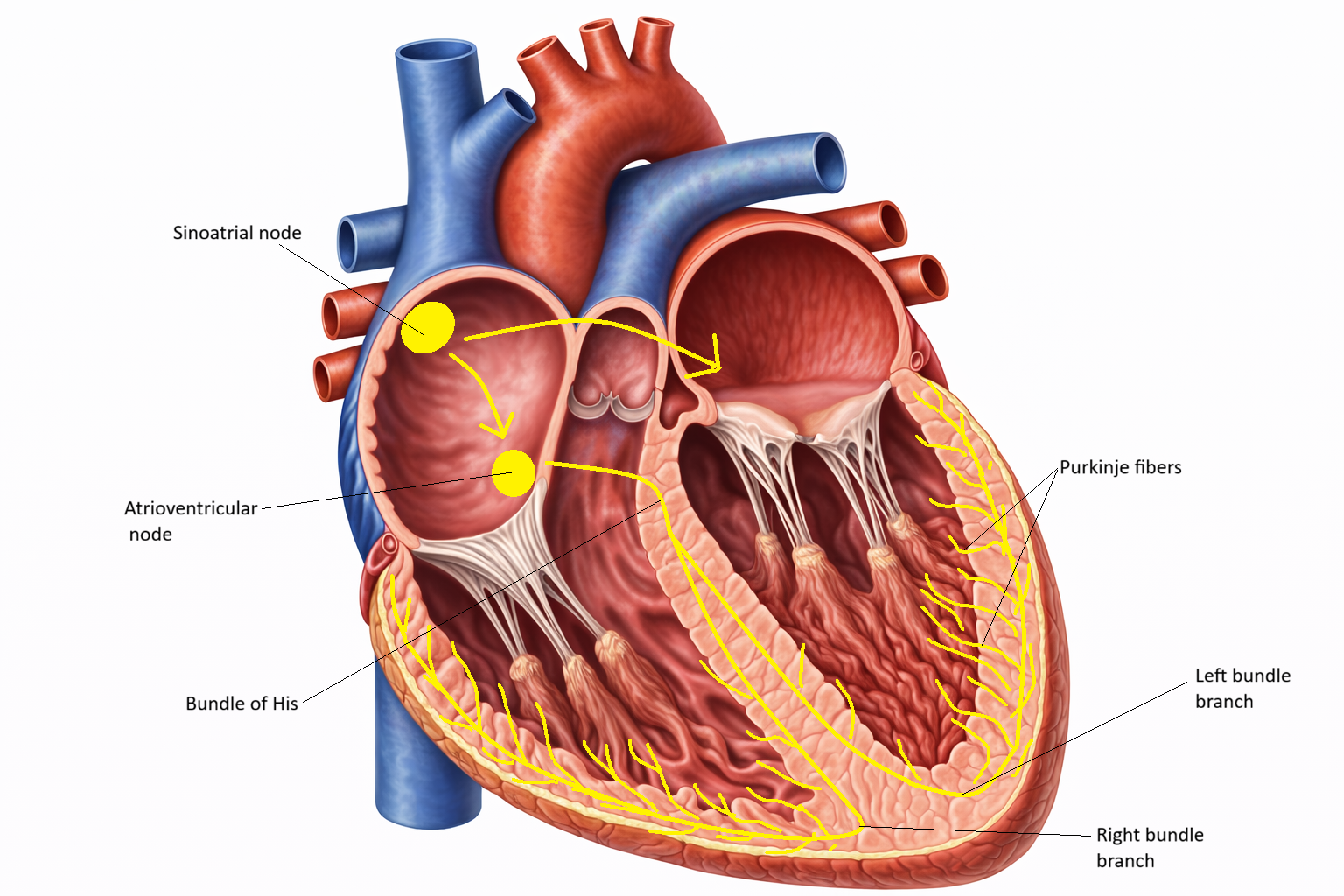
Image:Image: Cardiac conduction begins at the sinoatrial node, spreads across the atria to the atrioventricular node, then travels through the bundle of His, down the left and right bundle branches, and into the Purkinje fibres, coordinating ventricular contraction.
Beyond the Basics
Mechanisms of Arrhythmogenesis
Cardiac arrhythmias arise from disturbances in impulse formation, impulse conduction, or a combination of both. At the cellular level, these abnormalities reflect altered ion channel function, membrane instability, or disrupted electrical pathways within the myocardium.
Abnormal automaticity occurs when pacemaker cells fire at an inappropriate rate or when non-pacemaker cells develop spontaneous depolarising activity. The sinoatrial node may accelerate under increased sympathetic stimulation, producing sinus tachycardia, or subsidiary pacemakers may assume dominance if the primary pacemaker fails. Ischaemia can destabilise resting membrane potentials, allowing normally quiescent myocardial cells to generate spontaneous impulses.
Triggered activity results from afterdepolarisations, which are abnormal secondary depolarisations that occur during or after repolarisation. Early afterdepolarisations arise during the repolarisation phase and are commonly associated with prolonged QT intervals. Delayed afterdepolarisations occur after full repolarisation and are often linked to intracellular calcium overload. Electrolyte imbalance, drug toxicity, and certain inherited channelopathies increase susceptibility to these mechanisms.
Re-entry circuits are among the most common mechanisms underlying tachyarrhythmias. Re-entry requires a conduction pathway with two limbs that differ in conduction speed or refractory period. If one pathway conducts slowly while the other recovers excitability, an impulse can circle repeatedly, continuously reactivating tissue. This looping activation drives sustained tachyarrhythmias such as atrioventricular nodal re-entrant tachycardia and many forms of ventricular tachycardia.
Haemodynamic Consequences
The clinical impact of an arrhythmia depends not only on its electrical pattern but also on how it affects ventricular filling and effective forward flow. Very rapid rates shorten diastole, reducing time available for ventricular filling and lowering stroke volume. Very slow rates may fail to generate adequate cardiac output despite preserved filling. Loss of coordinated atrial contraction, as seen in atrial fibrillation, removes the contribution of atrial systole to ventricular filling, which can significantly reduce preload in individuals with stiff ventricles or diastolic dysfunction.
Uncoordinated ventricular activation further compromises efficiency. In ventricular tachycardia, rapid organised contractions limit filling time and reduce output. In ventricular fibrillation, chaotic electrical activity eliminates effective mechanical contraction altogether, resulting in immediate cessation of circulation. Reduced cardiac output manifests clinically as hypotension, dizziness, syncope, fatigue, or worsening heart failure, depending on severity and duration.
Atrial Versus Ventricular Arrhythmias
Atrial arrhythmias originate above the ventricles and primarily disrupt coordinated atrial contraction. While they may cause palpitations and haemodynamic compromise, their most significant long-term risk is thromboembolism, particularly in atrial fibrillation where blood stasis within the atria promotes clot formation. Although atrial arrhythmias can precipitate heart failure in susceptible individuals, they are less immediately lethal than malignant ventricular rhythms.
Ventricular arrhythmias arise within the ventricular myocardium and directly threaten systemic perfusion. Sustained ventricular tachycardia can markedly reduce cardiac output and may degenerate into ventricular fibrillation. Ventricular fibrillation produces complete loss of organised contraction and requires immediate defibrillation to restore circulation. The distinction between atrial and ventricular origin therefore has critical prognostic implications.
The Autonomic Nervous System and Arrhythmias
Autonomic tone significantly influences arrhythmia development. Sympathetic activation increases heart rate, shortens refractory periods, and enhances myocardial excitability, creating a substrate for ectopic activity and re-entry. Heightened sympathetic drive during stress, pain, hypoxia, or heart failure often precipitates tachyarrhythmias.
Parasympathetic stimulation, mediated primarily through the vagus nerve, slows sinoatrial firing and atrioventricular conduction. While increased vagal tone can protect against some tachyarrhythmias, abrupt shifts in autonomic balance may trigger others. Many clinically significant arrhythmias occur in settings of autonomic instability, where heightened sympathetic activity interacts with underlying structural or electrical vulnerability to initiate abnormal rhythms.
Clinical Connections
Cardiac arrhythmias are identified through careful ECG interpretation combined with clinical assessment. A 12-lead ECG provides information about rhythm origin, conduction intervals, QRS width, and repolarisation patterns, while continuous telemetry or ambulatory monitoring detects intermittent or paroxysmal events. Assessment of haemodynamic stability is equally important. Blood pressure, level of consciousness, chest pain, signs of shock, and evidence of heart failure determine urgency and guide immediate management. An irregular pulse with hypotension requires a different response from an asymptomatic rhythm detected incidentally on monitoring.
Clinical priorities differ depending on the rhythm pattern:
Narrow complex tachycardia with preserved blood pressure may be managed with rate control or vagal manoeuvres.
Atrial fibrillation with rapid ventricular response requires rate control and assessment of thromboembolic risk, often prompting anticoagulation.
Wide complex tachycardia with instability is treated as ventricular tachycardia until proven otherwise and may require urgent synchronised cardioversion.
Ventricular fibrillation or pulseless ventricular tachycardia requires immediate defibrillation and advanced life support.
Treatment strategies depend on the underlying mechanism and clinical impact. Rate control slows ventricular response without necessarily restoring sinus rhythm, while rhythm control aims to terminate the arrhythmia and maintain organised atrial activity. Antiarrhythmic drugs modify ion channel function but carry proarrhythmic risk, particularly in structurally abnormal hearts.
Catheter ablation, a minimally invasive procedure, targets focal triggers or re-entry pathways and can provide definitive therapy in selected tachyarrhythmias. In atrial fibrillation, anticoagulation reduces stroke risk by preventing thrombus formation in the left atrium. Ongoing rhythm surveillance remains essential because arrhythmia burden, recurrence, and haemodynamic tolerance can change over time, altering both prognosis and management decisions.
Concept Check
What are the three main mechanisms by which arrhythmias develop?
Why is loss of atrial kick clinically significant in atrial fibrillation?
How can re-entry circuits cause rapid tachyarrhythmias?
Why are ventricular arrhythmias more dangerous than atrial arrhythmias?
How does autonomic imbalance influence arrhythmia formation?