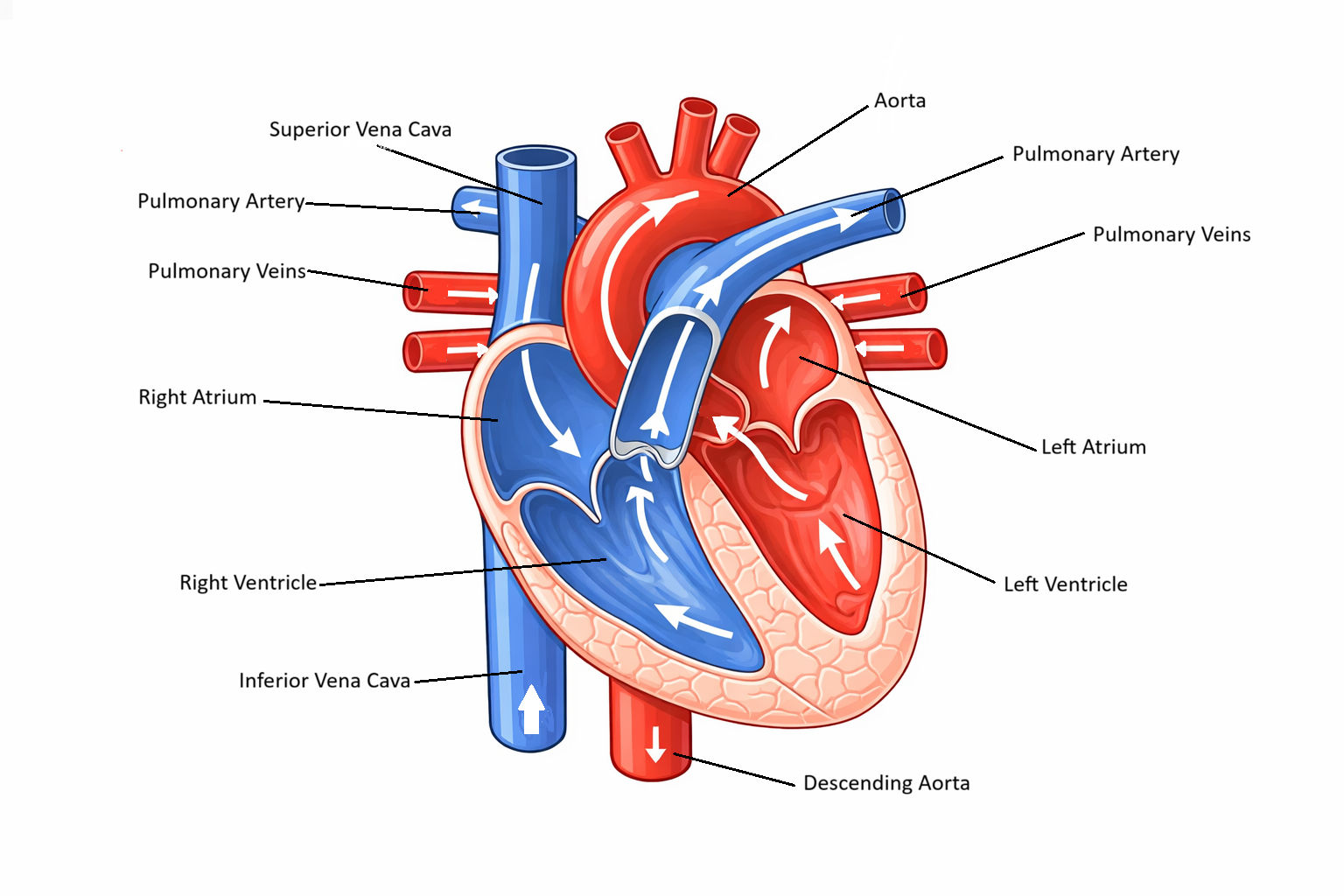
Blood Flow Through the Heart
Blood flow through the heart follows a precise and purposeful pathway that allows oxygen-poor blood to reach the lungs and oxygen-rich blood to supply the body. This journey depends on the coordinated contraction of the chambers and the proper functioning of the heart valves. Understanding the flow of blood is foundational to interpreting cardiovascular physiology, identifying abnormalities, and recognising conditions such as heart failure, valve disease, and pulmonary hypertension. For nursing students, mastery of this pathway is crucial for understanding vital signs, blood pressure, and the effects of common cardiac conditions.
What You Need to Know
Blood entering the heart from the body flows through the superior and inferior vena cava into the right atrium. From there, it moves through the tricuspid valve into the right ventricle. When the right ventricle contracts, blood is pushed through the pulmonary valve into the pulmonary artery and transported to the lungs. In the pulmonary capillaries, gas exchange occurs: carbon dioxide diffuses out of the blood and oxygen diffuses in.
Oxygenated blood returns to the heart via the pulmonary veins, the only veins that carry oxygen-rich blood, and enters the left atrium. It then flows through the mitral valve into the left ventricle, the most powerful chamber. When the left ventricle contracts, blood is ejected through the aortic valve and into the aorta, marking the beginning of systemic circulation. This pathway repeats with every heartbeat, maintaining continuous delivery of oxygen and nutrients to tissues.
Image: Deoxygenated blood flows from the body → superior and inferior vena cava → right atrium → right ventricle → pulmonary artery → lungs → pulmonary veins → left atrium → left ventricle → aorta → body.
Beyond the Basics
Pressure Gradients and Forward Flow
Blood does not move through the heart because it is “pushed forward,” but because pressure differences continuously form between chambers. Every valve in the heart opens or closes purely in response to these pressure gradients. During ventricular relaxation, pressure inside the ventricles drops rapidly, falling below atrial pressure. This creates a suction effect that draws blood from the atria into the ventricles, accounting for most ventricular filling at rest.
As filling continues, atrial and ventricular pressures gradually equalise and flow slows. Atrial contraction then provides a final pressure boost that tops up ventricular volume. This atrial kick becomes critical in situations where filling time is shortened, such as during tachycardia, or when the ventricle is stiff, as in hypertensive or hypertrophic heart disease. In these settings, loss of atrial contraction (for example, in atrial fibrillation) can cause dramatic drops in cardiac output.
Systolic Pressure Generation and Ventricular Ejection
Ventricular contraction does not immediately eject blood. Early systole is an isovolumetric phase where pressure rises sharply while all valves remain closed. Only once ventricular pressure exceeds the pressure in the aorta and pulmonary artery do the semilunar valves open and blood flow begins.
The strength and duration of ventricular contraction determine how steep this pressure rise is and how long ejection can be maintained. Stronger contraction creates a higher pressure gradient between ventricle and artery, driving faster and more complete emptying. As contraction wanes, ventricular pressure falls. When it drops below arterial pressure, blood attempts to flow backward, snapping the semilunar valves shut and producing S2. This closure is essential to maintain one-way flow and preserve forward circulation.
Venous Return as the True Driver of Cardiac Output
Although the heart generates pressure, it cannot pump blood that does not arrive. Venous return — the flow of blood back to the heart — is therefore the ultimate controller of cardiac output. The heart simply ejects what it receives.
Venous return is influenced by:
Blood volume
Venous tone (how constricted or relaxed veins are)
Skeletal muscle pumping
Intrathoracic pressure during breathing
When venous return increases, ventricular filling rises, stretching myocardial fibres and increasing stroke volume through the Frank–Starling mechanism. When venous return falls, such as during dehydration or haemorrhage, cardiac output falls even if the heart itself is healthy.
Preload and the Stretch–Force Relationship
Preload reflects how much the ventricle is filled before contraction begins. This filling stretches cardiac muscle fibres, positioning actin and myosin for optimal cross-bridge formation. Within normal limits, this stretch enhances contractile force, allowing the ventricle to eject more blood with the same amount of effort. However, excessive preload overstretches fibres, reducing efficiency and raising filling pressures. This is why fluid overload causes pulmonary oedema and congestion rather than improved cardiac output in heart failure — the ventricle is full, but it cannot pump effectively.
Afterload and Ejection Resistance
Afterload is the pressure the ventricle must overcome to eject blood. For the left ventricle, this is largely determined by systemic vascular resistance and aortic pressure. High afterload, such as in hypertension or aortic stenosis, means the ventricle must generate much higher pressure just to open the valve. This increases myocardial oxygen demand and reduces the amount of blood ejected with each beat. Over time, the ventricle thickens (hypertrophy) in an attempt to cope, but this eventually impairs relaxation and filling, linking afterload directly to diastolic dysfunction.
Dynamic Interaction of Flow, Pressure, and Resistance
Blood flow through the heart is the result of constantly shifting pressure gradients rather than fixed pump output. When venous return increases, preload rises, pressure gradients change, and cardiac output increases. When vascular resistance rises, afterload increases and output falls unless contractility compensates.
This dynamic balance allows the cardiovascular system to adapt rapidly to exercise, haemorrhage, posture changes, and illness. When any component fails, filling, contraction, or resistance, pressure gradients collapse and circulation becomes inefficient, explaining why heart failure, shock, and arrhythmias so profoundly impair organ perfusion.
Clinical Connections
When blood flow through the heart or lungs is impaired, predictable patterns of congestion and poor perfusion develop. Right-sided heart failure causes blood to back up into the systemic venous circulation, increasing venous pressure and pushing fluid into tissues. This explains why patients develop peripheral oedema, ascites, hepatic congestion, and jugular venous distension. Left-sided failure, in contrast, causes blood to accumulate in the pulmonary circulation, raising capillary pressures in the lungs and forcing fluid into the alveoli. The result is dyspnoea, orthopnoea, crackles, and impaired oxygen exchange.
Altered flow also explains many haemodynamic patterns:
Pulmonary hypertension increases resistance the right ventricle must pump against, leading to right ventricular hypertrophy and eventual failure
Systemic hypertension increases afterload on the left ventricle, reducing stroke volume and promoting left ventricular thickening and stiffness
Valve stenosis obstructs forward flow, causing pressure to build behind the valve and reducing cardiac output
Impaired flow through the coronary arteries also disrupts myocardial oxygen delivery. During ischaemia or infarction, the heart’s ability to generate pressure falls, reducing stroke volume and further worsening tissue perfusion. This creates a dangerous cycle in which poor flow leads to weaker contraction, which further reduces flow.
Low blood pressure may indicate reduced cardiac output, excessive afterload, or poor venous return. Oxygen saturation falls when pulmonary congestion or reduced cardiac output limits gas exchange. Heart sounds and murmurs reflect turbulent or obstructed flow across valves.
Pharmacological therapies are designed to modify flow and pressure:
Vasodilators reduce afterload, allowing blood to leave the heart more easily
Diuretics reduce preload, lowering congestion and pulmonary pressures
Inotropes increase contractile force, improving ejection when cardiac output is low
Any interruption to the normal flow pathway, whether from myocardial infarction, valve disease, pulmonary embolism, or structural heart defects, compromises oxygen delivery to organs. Rapid recognition of altered perfusion, congestion, or falling output is therefore central to preventing shock, respiratory failure, and multi-organ dysfunction.
Concept Check
Trace the exact pathway of blood from the vena cava to the aorta.
How does ventricular pressure influence the movement of blood during systole and diastole?
Why does left-sided heart failure lead to pulmonary symptoms?
What role do the pulmonary veins play in the cardiovascular system?
How does hypertension change the workload of the left ventricle?