The Adrenal Cortex
The adrenal cortex is the outer region of the adrenal gland and is essential for maintaining fluid balance, metabolic stability and the body’s ability to respond to stress. It produces three major classes of steroid hormones: mineralocorticoids, glucocorticoids and adrenal androgens; each arising from specialised zones within its layered structure. Because these hormones influence nearly every organ system, dysfunction of the adrenal cortex can cause life-threatening disturbances in blood pressure, glucose regulation, immunity and electrolyte balance. Understanding its zonal architecture and biochemical pathways is fundamental to interpreting adrenal disorders and endocrine feedback mechanisms.
What You Need to Know
The adrenal cortex is the outer region of the adrenal gland and is responsible for producing steroid hormones that regulate metabolism, fluid and electrolyte balance, stress responses, and aspects of sexual development. It is arranged into three distinct layers, each specialised for the synthesis of a particular class of hormones. This zonal organisation allows precise hormonal control despite all cortical hormones being derived from the same precursor, cholesterol.
Each zone of the adrenal cortex has a specific hormonal role:
The zona glomerulosa produces aldosterone
The zona fasciculata produces cortisol
The zona reticularis produces adrenal androgens, such as DHEA
Hormone synthesis within each zone depends on the presence of specific enzymes, which restrict hormone production to that layer. Aldosterone secretion is primarily regulated by angiotensin II and potassium levels as part of the renin–angiotensin–aldosterone system, while cortisol and adrenal androgen production are largely regulated by adrenocorticotropic hormone from the anterior pituitary. Cortisol plays a key role in glucose metabolism, immune modulation, and stress adaptation; aldosterone maintains sodium and potassium balance and therefore blood volume; and adrenal androgens contribute to pubertal development and act as precursors for sex hormones. The layered structure of the adrenal cortex ensures that these distinct physiological functions remain tightly regulated and responsive to changing demands.
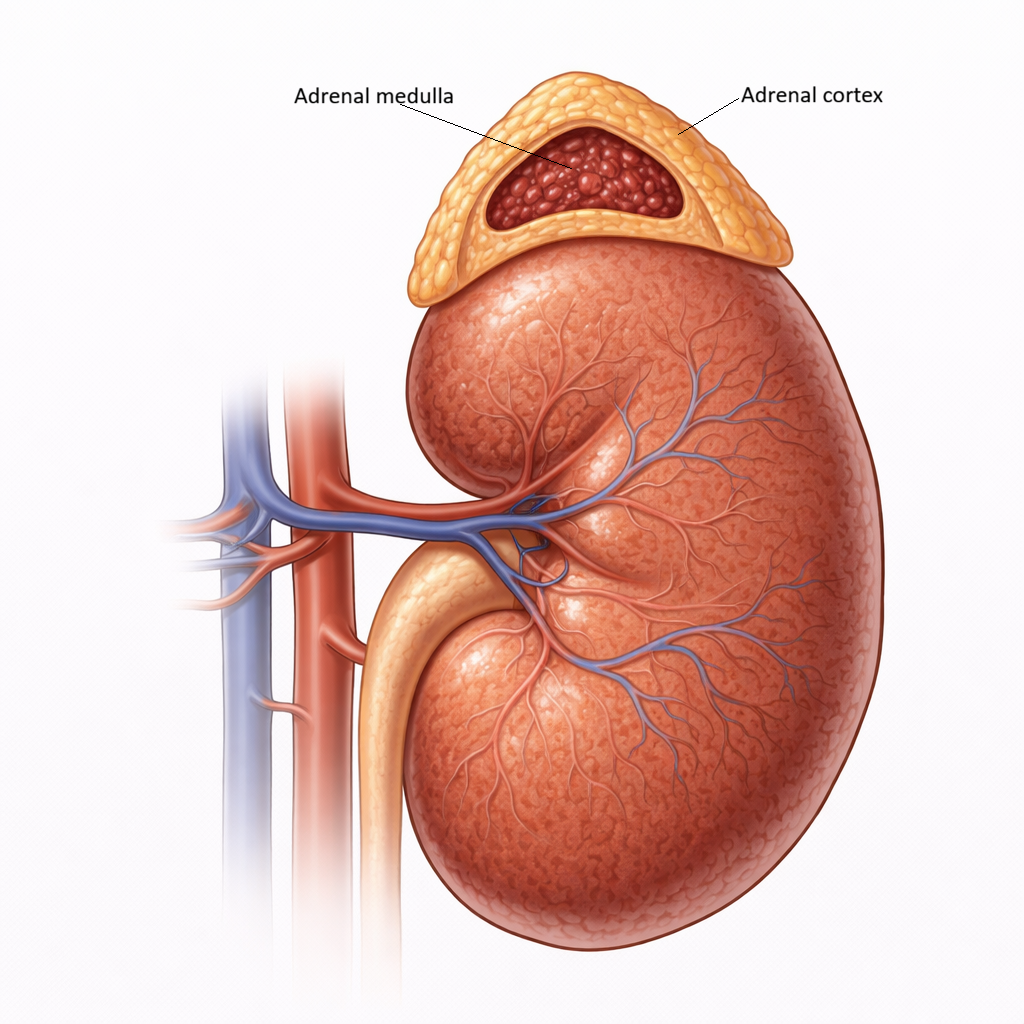
Image: The adrenal gland is composed of an outer cortex and an inner medulla. The cortex produces steroid hormones, while the medulla secretes catecholamines in response to sympathetic stimulation.
Beyond the Basics
Zonal organisation and cellular structure
The adrenal cortex is arranged into three concentric zones surrounding the adrenal medulla, with each layer specialised for the synthesis of a distinct class of steroid hormones. This zonal organisation is fundamental to adrenal function, as it allows the same precursor molecule, cholesterol, to be directed into different hormonal pathways depending on enzyme availability. The physical arrangement of the cortex reflects this functional separation, with each zone displaying characteristic cellular architecture linked to its hormone output.
The outermost zona glomerulosa is composed of cells arranged in rounded clusters, a structure that supports mineralocorticoid synthesis. These cells uniquely express aldosterone synthase, the enzyme required for the final steps of aldosterone production. Their enzyme profile prevents them from producing cortisol or androgens, ensuring that mineralocorticoid synthesis remains tightly restricted to this layer. This specificity is essential for precise regulation of sodium and potassium balance.
Beneath this lies the zona fasciculata, the thickest cortical layer and the primary site of glucocorticoid production. Its cells are arranged in long cords and contain abundant lipid droplets, reflecting the cholesterol-rich nature of steroid synthesis. The presence of extensive smooth endoplasmic reticulum and mitochondria with tubular cristae supports high-volume cortisol production under adrenocorticotropic hormone stimulation. The innermost zona reticularis contains smaller, more densely packed cells with darker cytoplasm, which produce adrenal androgens. Although these cells are also ACTH-responsive, their activity is further influenced by local paracrine signals, contributing to more subtle and variable hormone output.
Steroidogenesis and cholesterol handling
All adrenal cortical hormones are synthesised from cholesterol, making cholesterol availability and intracellular transport central to adrenal function. Cholesterol enters adrenal cortical cells either through uptake of circulating lipoproteins or mobilisation of stored intracellular lipid droplets. The critical regulatory step in steroidogenesis is the transport of cholesterol into the mitochondria, a process mediated by steroidogenic acute regulatory protein. This step determines the overall rate of hormone synthesis and is rapidly upregulated in response to hormonal stimulation.
Once inside the mitochondria, cholesterol undergoes enzymatic conversion into pregnenolone, which then serves as the precursor for all downstream adrenal steroids. Subsequent enzymatic steps occur within both the mitochondria and smooth endoplasmic reticulum, with the specific hormone produced determined by the enzymes present in each cortical zone. For example, the presence of aldosterone synthase in the zona glomerulosa enables mineralocorticoid synthesis, while the expression of 17α-hydroxylase in the zona fasciculata permits cortisol production. This zonal enzyme distribution ensures functional separation despite a shared substrate and prevents inappropriate hormone synthesis.
Regulation of cortisol secretion
Cortisol secretion is regulated by the hypothalamic–pituitary–adrenal axis. Corticotropin-releasing hormone from the hypothalamus stimulates the anterior pituitary to release ACTH, which in turn promotes cortisol synthesis and secretion from the zona fasciculata. In addition to stimulating hormone production, ACTH has a trophic effect on adrenal cortical cells, supporting their growth and structural integrity.
Cortisol secretion follows a circadian rhythm, with peak levels occurring in the early morning and progressively declining throughout the day. This rhythm supports daily metabolic demands, including glucose availability and vascular responsiveness. Cortisol secretion also increases in response to stressors such as illness, trauma, and hypoglycaemia. Through these mechanisms, cortisol helps maintain blood pressure, mobilise energy stores, and modulate inflammatory responses during periods of physiological challenge.
Regulation of aldosterone secretion
Aldosterone secretion is regulated primarily by the renin–angiotensin–aldosterone system rather than by pituitary control. Reduced renal perfusion, low sodium levels, or increased potassium concentration stimulate renin release from the kidneys, leading to formation of angiotensin II. Angiotensin II acts directly on the zona glomerulosa to stimulate aldosterone synthesis and release.
This regulatory pathway allows aldosterone secretion to respond rapidly to changes in fluid volume and electrolyte balance. Aldosterone increases sodium reabsorption and potassium excretion in the distal nephron, directly influencing blood volume and blood pressure. Although ACTH can transiently increase aldosterone secretion, it does not play a major role in long-term regulation. This relative independence from pituitary control ensures that electrolyte balance remains stable even when cortisol secretion fluctuates due to stress or circadian variation.
Adrenal androgen production
The zona reticularis produces weak androgens, primarily dehydroepiandrosterone and androstenedione. These hormones have limited androgenic activity on their own but serve as important precursors for more potent androgens and oestrogens in peripheral tissues. Their production is stimulated by ACTH, although regulation is less tightly linked to circadian rhythms compared with cortisol.
Adrenal androgens contribute to pubic and axillary hair development, libido, and broader metabolic effects, particularly in females. Unlike cortisol and aldosterone, adrenal androgen secretion declines with age, a process sometimes referred to as adrenopause. This age-related pattern highlights the distinct regulatory behaviour of the zona reticularis and reinforces the functional diversity within the adrenal cortex.
Clinical Connections
Disorders of the adrenal cortex produce recognisable clinical syndromes because each cortical zone produces hormones with distinct physiological roles. Changes in cortisol, aldosterone, or adrenal androgen levels tend to affect blood pressure, electrolytes, metabolism, and stress tolerance, meaning abnormalities are often first suspected through routine observations, blood tests, or unexplained systemic symptoms.
In clinical practice, adrenal cortical dysfunction is most often seen in the following patterns:
Excess or deficiency of cortisol
Abnormal aldosterone secretion affecting fluid and electrolyte balance
Altered androgen production
Excess cortisol secretion leads to Cushing’s syndrome, where prolonged exposure to high cortisol levels disrupts normal metabolism and tissue maintenance. Patients may develop central weight gain, thinning of the skin, easy bruising, muscle weakness, glucose intolerance, and hypertension. In contrast, cortisol deficiency, as seen in Addison’s disease, reduces the body’s ability to maintain vascular tone and respond to stress. This commonly presents as fatigue, weight loss, hypotension, hyponatraemia, and, in primary adrenal failure, skin hyperpigmentation due to elevated ACTH levels stimulating melanocyte receptors.
Disorders of aldosterone secretion primarily affect blood pressure and potassium balance. Hyperaldosteronism causes sodium retention and potassium loss, leading to hypertension and hypokalaemia that may be resistant to standard treatment. Aldosterone deficiency produces the opposite pattern, with salt wasting, dehydration, hypotension, and, in severe cases, circulatory collapse. These effects highlight the close link between mineralocorticoid function and cardiovascular stability.
Abnormal androgen production from the adrenal cortex may occur due to tumours or enzymatic defects in steroid synthesis. In these cases, excess androgen production can lead to virilisation or disrupted pubertal development, while reduced androgen output may contribute to low libido or delayed secondary sexual characteristics.
Concept Check
How does zonal enzyme expression determine which hormones are produced in each region of the adrenal cortex
Why is cholesterol essential for adrenal steroid hormone synthesis
How do ACTH and the renin–angiotensin–aldosterone system regulate different classes of adrenal cortical hormones
Why does cortisol follow a circadian secretion pattern
How can disorders of the adrenal cortex produce both metabolic and cardiovascular consequences