The Parathyroid Glands: Calcium Regulation, Hormone Secretion and Structural Specialisation
The parathyroid glands are small, often inconspicuous endocrine organs embedded on the posterior surface of the thyroid gland, yet they play a disproportionately large role in maintaining physiological stability. Their primary function is the regulation of calcium and phosphate balance—processes essential for neuromuscular function, bone integrity, blood clotting and cellular signalling. Through the secretion of parathyroid hormone, the parathyroid glands act on bone, kidneys and the gastrointestinal tract to ensure precise control of circulating calcium levels. Understanding their structure provides insight into disorders such as hypocalcaemia, hyperparathyroidism and disturbances of bone metabolism.
What You Need to Know
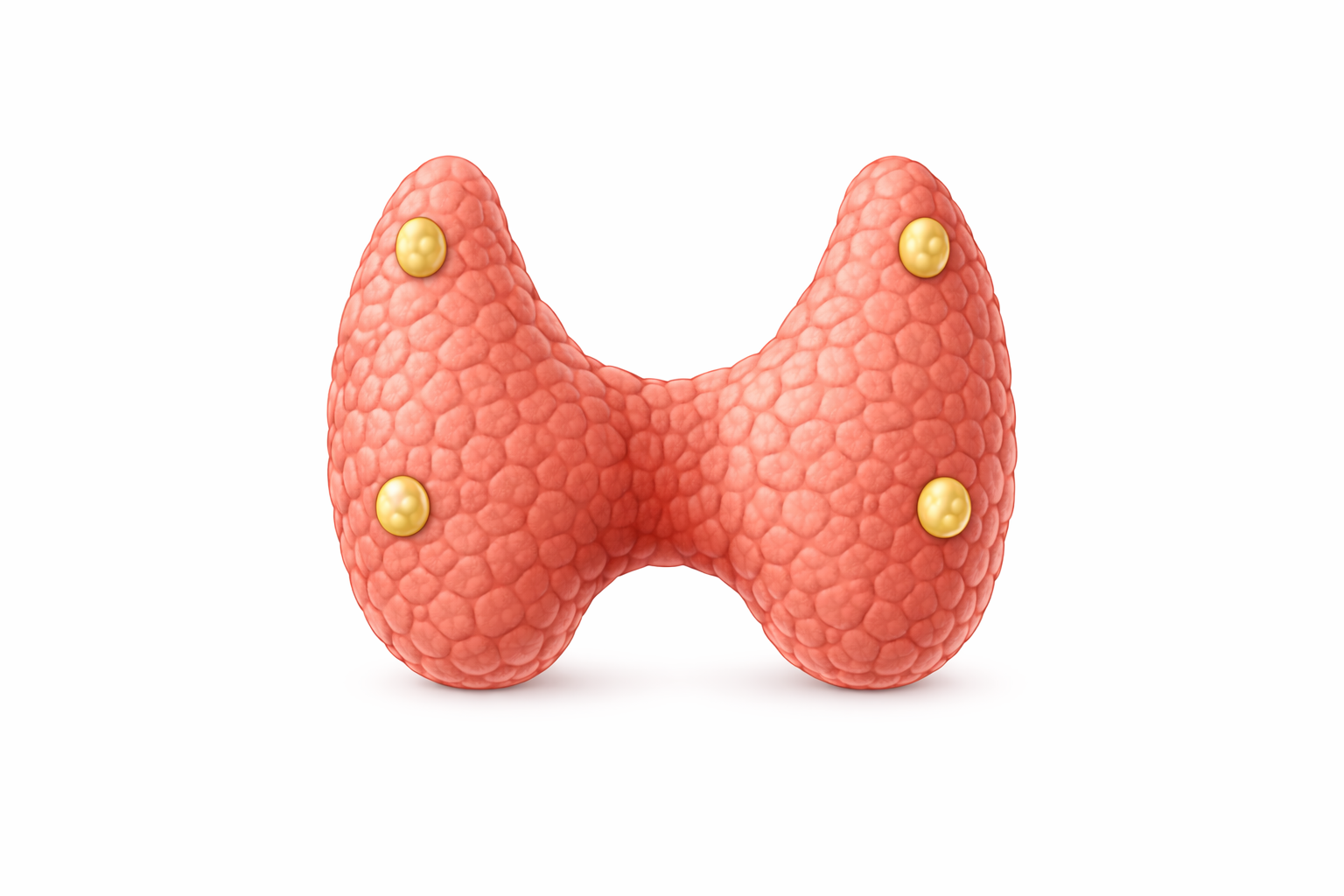
The parathyroid glands are small endocrine glands, most commonly four in number, located on the posterior surface of the thyroid, although variation in number and position is common. Their primary function is to maintain stable blood calcium levels, which are essential for normal neuromuscular activity, cardiac conduction, blood clotting, and intracellular signalling. Because calcium balance is so tightly regulated, even mild disturbances in parathyroid function can lead to widespread physiological effects.
Parathyroid hormone is synthesised and secreted by chief cells in direct response to falling blood calcium levels detected by calcium-sensing receptors on the cell surface. Unlike many endocrine hormones, parathyroid hormone is not stored in large amounts but is released rapidly as calcium levels change, allowing tight moment-to-moment control. The hormone acts across multiple organ systems to restore calcium balance, coordinating bone, renal, and gastrointestinal responses.
The key actions of parathyroid hormone include:
Raising blood calcium through effects on bone, kidneys, and the gastrointestinal tract
Increasing renal calcium reabsorption while reducing phosphate reabsorption
Promoting activation of vitamin D to enhance intestinal calcium absorption
In addition to chief cells, the parathyroid glands contain oxyphil cells, which are present in smaller numbers and tend to increase with age. Their exact function remains unclear, but they are thought to represent a different functional or metabolic state of parathyroid tissue rather than a primary hormone-secreting population. Together, the cellular composition and rapid hormone responsiveness of the parathyroid glands reflect their specialised role in maintaining calcium homeostasis.
Image: Parathyroid glands on the posterior surface of the thyroid gland.
Beyond the Basics
Microanatomy and cellular composition
Each parathyroid gland is composed of tightly packed cords of cells separated by a fine connective tissue stroma that contains an extensive capillary network. This dense vascular supply is required due to the gland’s role in rapid, moment-to-moment regulation of calcium levels, allowing parathyroid hormone to enter the circulation almost immediately after secretion. Unlike many endocrine glands that store hormone for later release, the parathyroid glands rely on continuous synthesis and rapid responsiveness.
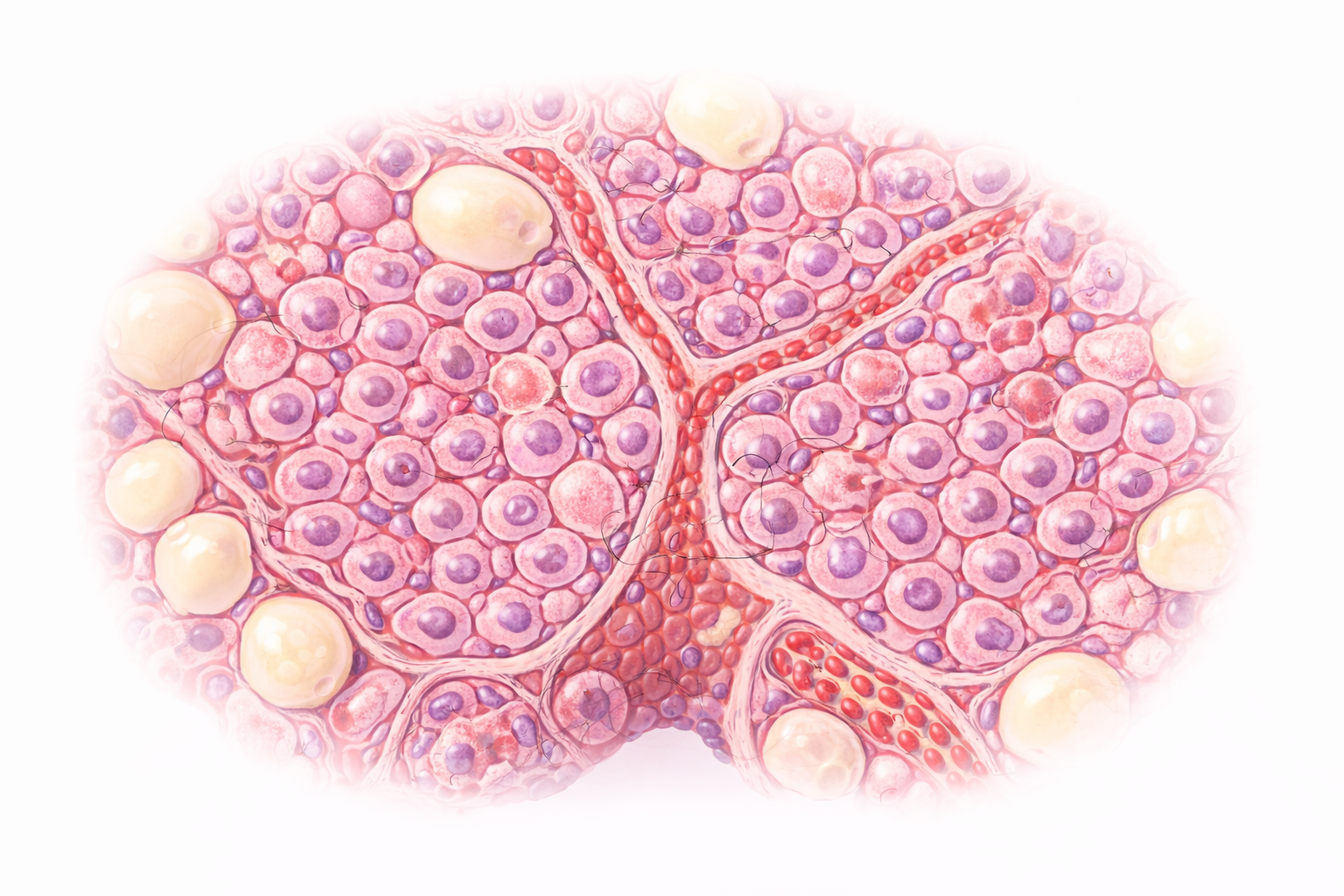
Chief cells are the dominant cell type and are responsible for synthesising and secreting parathyroid hormone. Their cytoplasm contains abundant rough endoplasmic reticulum and prominent secretory granules, consistent with ongoing protein hormone production. Oxyphil cells are larger, with eosinophilic, mitochondria-rich cytoplasm and far fewer secretory granules. Although their exact function remains uncertain, they appear to have reduced secretory activity and increase in number with age and in certain disease states, including chronic kidney disease. Their presence highlights functional heterogeneity within the gland rather than a simple uniform population of hormone-secreting cells.
Image: The parathyroid gland contains densely packed chief cells, which produce parathyroid hormone, alongside larger oxyphil cells and a rich capillary network. Scattered adipose tissue becomes more prominent with age.
Calcium-sensing and feedback control
Chief cells regulate parathyroid hormone secretion through calcium-sensing receptors located on their cell membrane. These receptors allow the cells to detect very small changes in extracellular calcium concentration, making calcium itself the primary regulator of hormone release. When blood calcium levels fall, the inhibitory signal on the chief cells is removed, leading to immediate parathyroid hormone secretion. When calcium levels rise, hormone release is suppressed.
This receptor-based control system allows calcium levels to be maintained within an exceptionally narrow physiological range. Such precision is essential because calcium plays a critical role in neuromuscular excitability, cardiac conduction, and intracellular signalling. Rather than relying on delayed hormonal cascades, the parathyroid glands respond directly and rapidly to changes in calcium concentration, providing tight homeostatic control.
Actions of parathyroid hormone on bone
Parathyroid hormone influences bone metabolism through its effects on the balance between bone formation and resorption. With sustained elevation, parathyroid hormone promotes osteoclast-mediated bone resorption indirectly, increasing the release of calcium and phosphate into the bloodstream. Over time, this leads to reduced bone mineral density and characteristic skeletal changes seen in chronic hyperparathyroidism.
In contrast, intermittent exposure to parathyroid hormone has a different effect. Short, transient increases stimulate osteoblast activity and promote bone formation rather than resorption. This dual action explains why parathyroid hormone analogues can be used therapeutically in osteoporosis, while chronic excess hormone exposure weakens the skeleton. Understanding this time-dependent effect is key to linking hormone physiology with both disease patterns and treatment strategies.
Renal and gastrointestinal integration
The kidneys play a central role in parathyroid hormone action and calcium balance. Parathyroid hormone increases calcium reabsorption in the distal nephron, reducing urinary calcium loss, while simultaneously decreasing phosphate reabsorption, leading to increased phosphate excretion. This reciprocal handling of calcium and phosphate helps prevent precipitation of calcium–phosphate complexes in the circulation.
Parathyroid hormone also stimulates the renal enzyme responsible for converting inactive vitamin D into its active form, calcitriol. Calcitriol then acts on the gastrointestinal tract to increase absorption of dietary calcium and phosphate. Through this pathway, parathyroid hormone indirectly links renal function to intestinal calcium uptake, completing a coordinated, multi-organ system that maintains calcium homeostasis across bone, kidneys, and gut.
Embryological origin and anatomical variability
The parathyroid glands develop from the third and fourth pharyngeal pouches during embryogenesis. Their migration during development explains why their final anatomical position is highly variable in adults. While most parathyroid glands are located on the posterior surface of the thyroid, ectopic tissue may be found elsewhere in the neck, within the thymus, or in the mediastinum.
This variability has important clinical implications, particularly during parathyroid surgery or when investigating persistent hormone excess. Difficulty locating all functioning parathyroid tissue can complicate both diagnosis and treatment. Understanding the embryological origin of the glands provides a framework for anticipating atypical locations and interpreting imaging findings when standard anatomical expectations are not met.
Clinical Connections
Disorders of the parathyroid glands most often present through abnormalities in calcium levels, rather than obvious gland-related symptoms. Because parathyroid hormone has direct effects on bone, kidneys, and neuromuscular function, changes in its secretion tend to produce recognisable patterns across laboratory results, imaging, and clinical features. Linking these findings back to parathyroid physiology helps make sense of otherwise broad or nonspecific presentations.
In clinical practice, parathyroid dysfunction is most commonly seen in the following patterns:
Excess parathyroid hormone with elevated blood calcium
Insufficient parathyroid hormone with low blood calcium
Hyperparathyroidism, whether primary, secondary, or tertiary, results in excess parathyroid hormone secretion and, in many cases, hypercalcaemia. Patients may present with bone pain, recurrent kidney stones, abdominal discomfort, or changes in concentration and mood. Over time, sustained hormone excess increases bone resorption, leading to reduced bone density and characteristic skeletal changes that may be visible on imaging. Renal involvement is also common, reflecting increased calcium filtration and stone formation.
Hypoparathyroidism leads to reduced parathyroid hormone activity and hypocalcaemia, most commonly following neck surgery or due to autoimmune destruction of the glands. Low calcium levels increase neuromuscular excitability, producing symptoms such as perioral tingling, muscle cramps, tetany, and, in more severe cases, cardiac arrhythmias. These features often develop quickly and correlate closely with biochemical findings, highlighting the tight relationship between calcium levels and clinical presentation.
Concept Check
How do the calcium-sensing receptors of chief cells regulate PTH secretion
Why does intermittent PTH exposure stimulate bone formation while chronic elevation promotes bone resorption
How does PTH influence renal handling of calcium and phosphate
Why is vitamin D activation essential for maintaining calcium homeostasis
How do developmental pathways contribute to anatomical variation in parathyroid gland location