The Anterior Pituitary: Hormone Synthesis, Regulation and Systemic Influence
The anterior pituitary, or adenohypophysis, is one of the most influential endocrine glands in the body. Acting under the control of the hypothalamus, it synthesises and secretes a range of hormones that regulate growth, metabolism, reproduction, stress responses and thyroid function. Because its hormones act on multiple peripheral endocrine glands, the anterior pituitary is a central regulator within several feedback loops. Alterations in its structure or function produce widespread and sometimes profound physiological changes, making it essential to understand its anatomy and regulatory mechanisms.
What You Need to Know
The anterior pituitary is the central hormonal relay between the hypothalamus and peripheral endocrine organs. It is composed of glandular epithelial tissue derived from Rathke’s pouch and is uniquely regulated by hypothalamic releasing and inhibiting hormones delivered through the hypophyseal portal system. This vascular arrangement allows small changes in hypothalamic signalling to exert precise and rapid control over pituitary hormone secretion without dilution in the systemic circulation.
Hormones produced by the anterior pituitary either act directly on target tissues or stimulate other endocrine glands to release their own hormones. Through this hierarchical organisation, the anterior pituitary coordinates growth, metabolism, stress responses, reproduction and lactation. Its output is continuously adjusted through negative feedback loops, ensuring hormonal stability while allowing rapid adaptation to physiological demand.
The six major anterior pituitary hormones and their primary roles include:
Growth hormone, which stimulates tissue growth and metabolic regulation, largely through IGF-1
Thyroid-stimulating hormone, which regulates thyroid hormone production and metabolic rate
Adrenocorticotropic hormone, which drives cortisol secretion from the adrenal cortex
Prolactin, which promotes lactation and has modulatory effects on reproductive function
Follicle-stimulating hormone, which supports gametogenesis in ovaries and testes
Luteinising hormone, which triggers ovulation and stimulates sex steroid production
Secretion of these hormones is governed by tightly regulated feedback loops involving cortisol, thyroid hormones, sex steroids and insulin-like growth factor-1. Disruption at any level of the hypothalamic–pituitary–target gland axis can therefore produce characteristic laboratory patterns and clinical syndromes. Understanding these relationships is essential for distinguishing primary gland failure from secondary or tertiary endocrine disorders and for accurately interpreting pituitary hormone results in clinical practice.
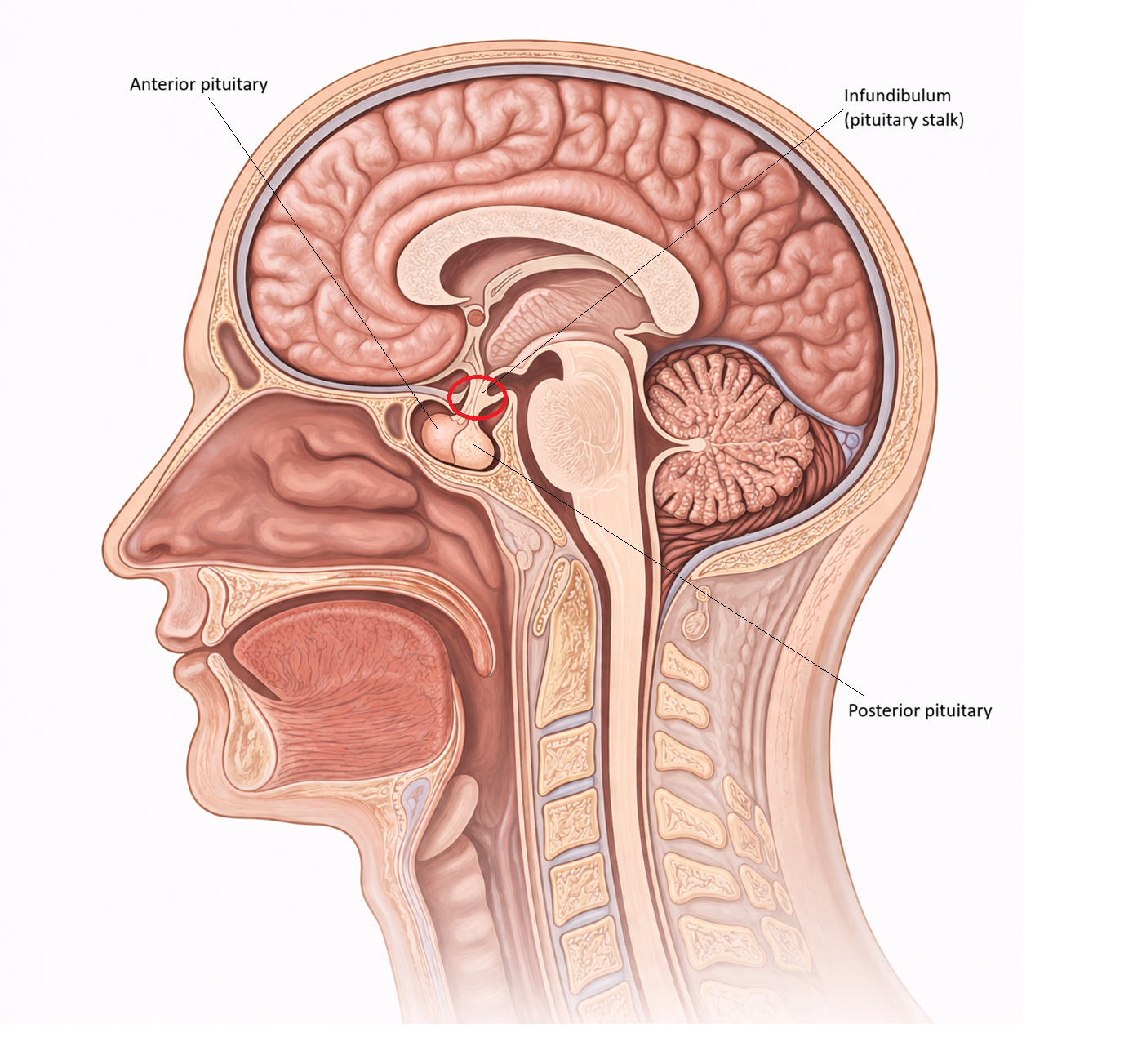
Image: The anterior pituitary is the glandular component of the pituitary that synthesises and releases key hormones involved in growth, metabolism, reproduction, and stress responses.
Beyond the Basics
Cellular Architecture
The anterior pituitary is composed of several highly specialised endocrine cell populations, each responsible for synthesising and secreting a distinct hormone or group of hormones. Somatotrophs are the most abundant cell type and secrete growth hormone in response to growth hormone–releasing hormone, with secretion inhibited by somatostatin. Their activity integrates hypothalamic signalling with metabolic cues such as sleep, nutrition and exercise.
Thyrotrophs produce thyroid-stimulating hormone under the control of thyrotropin-releasing hormone and are tightly regulated by circulating thyroid hormone levels. Corticotrophs synthesise adrenocorticotropic hormone, which is derived from the precursor molecule pro-opiomelanocortin and gives rise to several biologically active peptides. These cells respond to corticotropin-releasing hormone and are central to activation of the stress response through adrenal cortisol secretion.
Gonadotrophs produce follicle-stimulating hormone and luteinising hormone in response to pulsatile gonadotropin-releasing hormone. The frequency and amplitude of GnRH pulses determine the relative secretion of FSH and LH, allowing fine control of reproductive function. Lactotrophs secrete prolactin and are unique among anterior pituitary cells because their dominant regulatory input is inhibitory rather than stimulatory. Dopamine from the hypothalamus suppresses prolactin release, which explains why disruption of hypothalamic input often results in elevated prolactin levels. This diversity of cell types allows the anterior pituitary to regulate multiple physiological systems simultaneously.
Regulation Through the Hypophyseal Portal System
The hypophyseal portal system is essential for precise hypothalamic control of anterior pituitary secretion. Releasing and inhibiting hormones are secreted into the primary capillary plexus at the median eminence, then transported via portal veins to the secondary capillary plexus within the anterior pituitary. This direct vascular connection ensures that hypothalamic hormones reach their target cells rapidly and at effective concentrations.
Because this system bypasses the systemic circulation, very small amounts of hypothalamic hormone can exert powerful regulatory effects. Disruption of the portal circulation, such as through pituitary stalk compression or injury, reduces delivery of releasing hormones and often leads to combined anterior pituitary hormone deficiencies. In contrast, prolactin levels typically rise under these conditions due to loss of dopaminergic inhibition.
Feedback Loops
Each anterior pituitary hormone operates within a negative feedback loop that stabilises endocrine output and prevents excessive hormone secretion. Rising thyroid hormone levels suppress further TSH release, while increasing cortisol concentrations inhibit ACTH secretion at both pituitary and hypothalamic levels. Sex steroids similarly regulate follicle-stimulating hormone and luteinising hormone release to maintain reproductive balance.
Growth hormone regulation is more complex and involves multiple feedback pathways. Insulin-like growth factor-1, produced primarily by the liver, suppresses growth hormone secretion, while metabolic factors such as hypoglycaemia, exercise and sleep stimulate release. Prolactin differs from other pituitary hormones because it lacks a classical peripheral endocrine feedback loop. Instead, its secretion is restrained primarily by hypothalamic dopamine, making it particularly sensitive to disruptions in hypothalamic control.
Integration With Stress, Reproduction and Growth
The anterior pituitary sits at the centre of several major physiological axes. Adrenocorticotropic hormone drives cortisol production, which is essential for stress adaptation, immune modulation and metabolic stability. Follicle-stimulating hormone and luteinising hormone regulate gametogenesis and sex steroid production, linking pituitary function directly to fertility and sexual development.
Growth hormone influences linear growth during childhood and plays a key role in adult metabolism by promoting protein synthesis, lipid utilisation and glucose regulation. Through coordinated control of these axes, the anterior pituitary integrates environmental signals, internal physiological state and long-term adaptive responses. This structural and functional organisation underpins its role as a master regulator of endocrine function rather than a simple hormone relay station.
Clinical Connections
Disorders of the anterior pituitary produce predictable but far-reaching clinical syndromes because this gland sits upstream of multiple endocrine axes. Pituitary adenomas are the most common cause of anterior pituitary dysfunction. These tumours may be hormonally active, secreting excess hormone, or non-functioning, causing disease through compression of normal pituitary tissue and disruption of hypothalamic input.
Hormone-secreting adenomas give rise to characteristic clinical patterns depending on the cell type involved:
• Prolactin-secreting adenomas cause hyperprolactinaemia, leading to menstrual irregularities, galactorrhoea, infertility and hypogonadism
• ACTH-secreting adenomas result in Cushing’s disease, characterised by cortisol excess, central adiposity, muscle weakness, hypertension and metabolic disturbance
• Growth hormone–secreting adenomas cause acromegaly in adults or gigantism in children, with progressive soft tissue overgrowth, insulin resistance and cardiovascular complications
Non-functioning adenomas often present later, once mass effect develops. Compression of surrounding pituitary tissue reduces hormone output, while optic chiasm compression may cause bitemporal hemianopia. Headache, visual disturbance and gradual endocrine failure are common presenting features.
Damage to the pituitary gland or pituitary stalk from surgery, trauma, radiotherapy, infarction or infiltrative disease can result in panhypopituitarism. In this setting, multiple hormonal axes are affected simultaneously, producing combined deficiencies of cortisol, thyroid hormone, sex steroids and growth hormone. Prolactin levels may be normal or elevated due to loss of dopaminergic inhibition, providing an important diagnostic clue to stalk involvement.
Abnormal peripheral hormone levels must always be interpreted alongside pituitary hormones to distinguish primary gland failure from secondary pituitary disease. Because anterior pituitary disorders affect growth, metabolism, reproduction and stress responses, early recognition has significant implications for long-term morbidity and quality of life.
Concept Check
Why does the anterior pituitary require the hypophyseal portal system for effective regulation
How do feedback loops involving thyroid hormone, cortisol and sex steroids maintain pituitary stability
Why is prolactin primarily regulated by inhibition rather than stimulation
How can a pituitary adenoma lead to both hormone excess and hormone deficiency
Why do disruptions of the pituitary stalk often result in hyperprolactinaemia