The Hypothalamus: Neural–Endocrine Integration and Hormonal Control
The hypothalamus is the central coordinating hub that links the nervous system to the endocrine system. Situated at the base of the brain and forming the floor of the third ventricle, it integrates neural information from across the body and converts it into hormonal signals. These signals regulate temperature, hunger, thirst, circadian rhythms, stress responses, reproductive function and fluid balance. The hypothalamus exerts its influence primarily through releasing and inhibiting hormones that act on the anterior pituitary, as well as through direct secretion of hormones stored in the posterior pituitary. Because of its extensive regulatory role, disturbances in hypothalamic function can have widespread physiological consequences.
What You Need to Know
The hypothalamus is the primary link between the nervous system and the endocrine system, translating neural signals into hormonal responses. It continuously integrates information about stress, temperature, light–dark cycles, nutritional status, fluid balance and emotional state, then adjusts endocrine output to maintain homeostasis. Rather than acting as a passive relay, the hypothalamus actively coordinates hormonal timing, amplitude and feedback sensitivity across multiple endocrine axes.
Hormonal control of the anterior pituitary occurs through the hypophyseal portal system, a specialised vascular network that allows hypothalamic releasing and inhibiting hormones to reach the pituitary in high concentration without dilution in the systemic circulation. This arrangement enables precise regulation of pituitary hormone secretion in response to rapidly changing physiological demands.
The hypothalamus regulates pituitary function through several key hormones, including:
Releasing hormones, such as thyrotropin-releasing hormone, corticotropin-releasing hormone, gonadotropin-releasing hormone and growth hormone-releasing hormone, which stimulate anterior pituitary secretion
Inhibiting hormones, including somatostatin and dopamine, which suppress release of growth hormone and prolactin respectively
In addition to its control over the anterior pituitary, the hypothalamus directly synthesises antidiuretic hormone and oxytocin within the supraoptic and paraventricular nuclei. These hormones are transported along axons to the posterior pituitary, where they are stored and released into the circulation in response to neural signals. This dual mechanism allows the hypothalamus to deliver both rapid neural responses and sustained endocrine effects, positioning it as the central coordinator of hormonal regulation across the body.
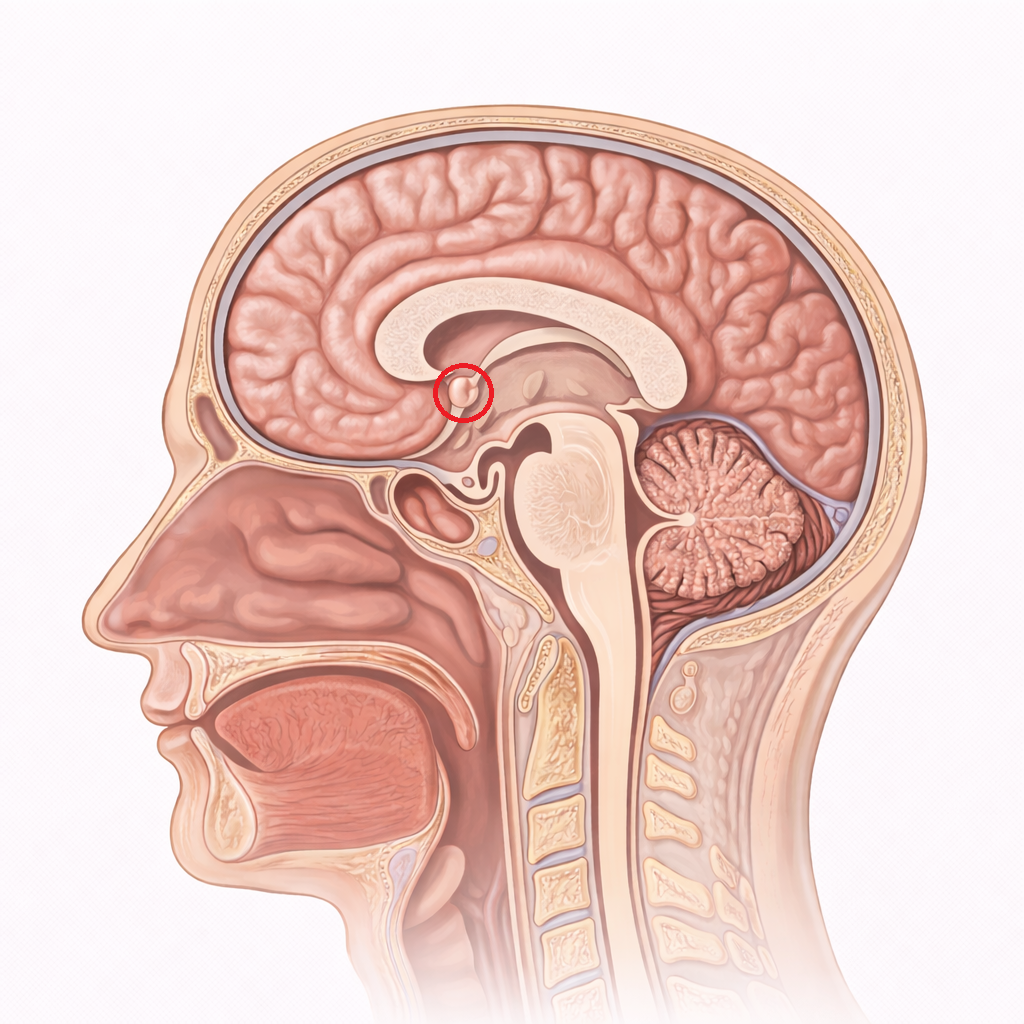
Image: The hypothalamus is located deep within the brain, forming the floor of the third ventricle and lying inferior to the thalamus. It acts as a key link between the nervous and endocrine systems, regulating processes such as temperature, appetite, and hormone release.
Beyond the Basics
Anatomical Nuclei and Their Functions
The hypothalamus is organised into multiple anatomically and functionally distinct nuclei, each responsible for regulating specific endocrine, autonomic and behavioural processes. These nuclei act as specialised processing centres rather than isolated units, allowing the hypothalamus to integrate diverse physiological signals and generate coordinated hormonal responses.
The paraventricular nucleus plays a central role in oxytocin synthesis and also contributes significantly to corticotropin-releasing hormone production, linking stress perception with activation of the hypothalamic–pituitary–adrenal axis. The supraoptic nucleus is the principal source of antidiuretic hormone, making it essential for long-term regulation of plasma osmolality and fluid balance. The arcuate nucleus is critically involved in metabolic regulation and appetite control and serves as a major source of dopamine, which exerts tonic inhibitory control over prolactin secretion in the anterior pituitary.
Other hypothalamic nuclei participate in thermoregulation, circadian rhythm control, reproductive hormone release and autonomic nervous system regulation. Through extensive connections with the brainstem, limbic system and higher cortical centres, these nuclei integrate emotional, metabolic and sensory inputs, ensuring that endocrine output reflects both internal physiological state and external environmental conditions.
Hypophyseal Portal System
The hypophyseal portal system is one of the most specialised vascular arrangements in the body and is fundamental to hypothalamic control of the anterior pituitary. It consists of a primary capillary plexus in the median eminence, which collects hypothalamic releasing and inhibiting hormones, and a secondary capillary plexus in the anterior pituitary, where these hormones exert their effects.
This two-capillary network allows very small quantities of hypothalamic hormones to reach the pituitary at high concentration without dilution in the systemic circulation. As a result, subtle changes in hypothalamic signalling can produce large downstream effects on thyroid function, adrenal activity, reproductive cycles and growth regulation. Disruption of the portal system, whether through trauma, vascular compromise or mass lesions, can therefore cause pituitary hormone deficiencies even when pituitary tissue remains structurally intact.
Neurosecretion and Posterior Pituitary Regulation
In contrast to the anterior pituitary, the posterior pituitary functions as a neurosecretory extension of the hypothalamus rather than an independent endocrine gland. Oxytocin and antidiuretic hormone are synthesised in the cell bodies of hypothalamic neurons and transported along axons to nerve terminals in the posterior pituitary, where they are stored until release.
Hormone secretion from the posterior pituitary is triggered directly by neural activity. For example, increases in plasma osmolality are detected by hypothalamic osmoreceptors, leading to rapid release of antidiuretic hormone and promotion of renal water retention. Similarly, sensory input from cervical stretch during labour or nipple stimulation during breastfeeding activates oxytocin release, generating positive feedback loops that are essential for effective parturition and milk ejection.
Integration of Neural and Endocrine Function
The hypothalamus occupies a unique position at the interface between neural processing and endocrine regulation. Its connections with the limbic system allow emotional states such as stress, fear and bonding to influence hormone secretion, explaining why psychological factors can profoundly affect appetite, fertility, sleep and immune function.
Temperature regulation provides another example of this integration. Hypothalamic thermosensitive neurons monitor blood temperature and coordinate behavioural responses, autonomic adjustments and hormonal changes to maintain thermal homeostasis. Through this continuous integration of sensory, emotional and physiological information, the hypothalamus ensures that endocrine signalling remains adaptive, context-sensitive and tightly aligned with the body’s immediate and long-term needs.
Clinical Connections
Damage to the hypothalamus often produces complex and multisystem endocrine disorders because a single lesion can disrupt multiple hormonal axes simultaneously. Unlike isolated pituitary disease, hypothalamic dysfunction commonly presents with mixed patterns of hormone deficiency or dysregulation, reflecting impaired releasing hormone control rather than failure of the target endocrine glands themselves. This makes clinical interpretation more challenging and highlights the importance of understanding hypothalamic anatomy and function.
Injury to specific hypothalamic nuclei produces predictable clinical syndromes. Damage to the supraoptic or paraventricular nuclei impairs antidiuretic hormone synthesis, resulting in central diabetes insipidus with polyuria, polydipsia and hypernatraemia. Lesions involving the arcuate nucleus disrupt appetite regulation and dopaminergic inhibition of prolactin, leading to marked weight changes, hyperprolactinaemia or reproductive dysfunction. Because these nuclei also integrate metabolic and hormonal signals, symptoms often extend beyond a single endocrine axis.
Common clinical consequences of hypothalamic damage include:
Diabetes insipidus due to impaired ADH synthesis and release
Disordered appetite and weight regulation from arcuate nucleus dysfunction
Combined pituitary hormone deficiencies caused by reduced releasing hormone production
Apparent pituitary failure resulting from disruption of the hypophyseal portal system
Hypothalamic tumours, traumatic brain injury, infections, vascular insults and infiltrative diseases such as sarcoidosis can all impair hypothalamic signalling. When the hypophyseal portal system is affected, anterior pituitary hormone secretion may be globally reduced despite structurally intact pituitary tissue. This pattern contrasts with primary pituitary disease, where hormone loss often follows a more predictable sequence.
Concept Check
Why is the hypophyseal portal system essential for effective communication between the hypothalamus and anterior pituitary
How does the hypothalamus integrate neural and endocrine signals to regulate homeostasis
Why can a lesion in the hypothalamus produce multiple hormonal deficiencies rather than a single endocrine disorder
How does hypothalamic control of the posterior pituitary differ from its control of the anterior pituitary
Which hypothalamic nuclei are primarily responsible for producing oxytocin and antidiuretic hormone