The Posterior Pituitary: Neurosecretion, Hormone Release and Fluid Balance
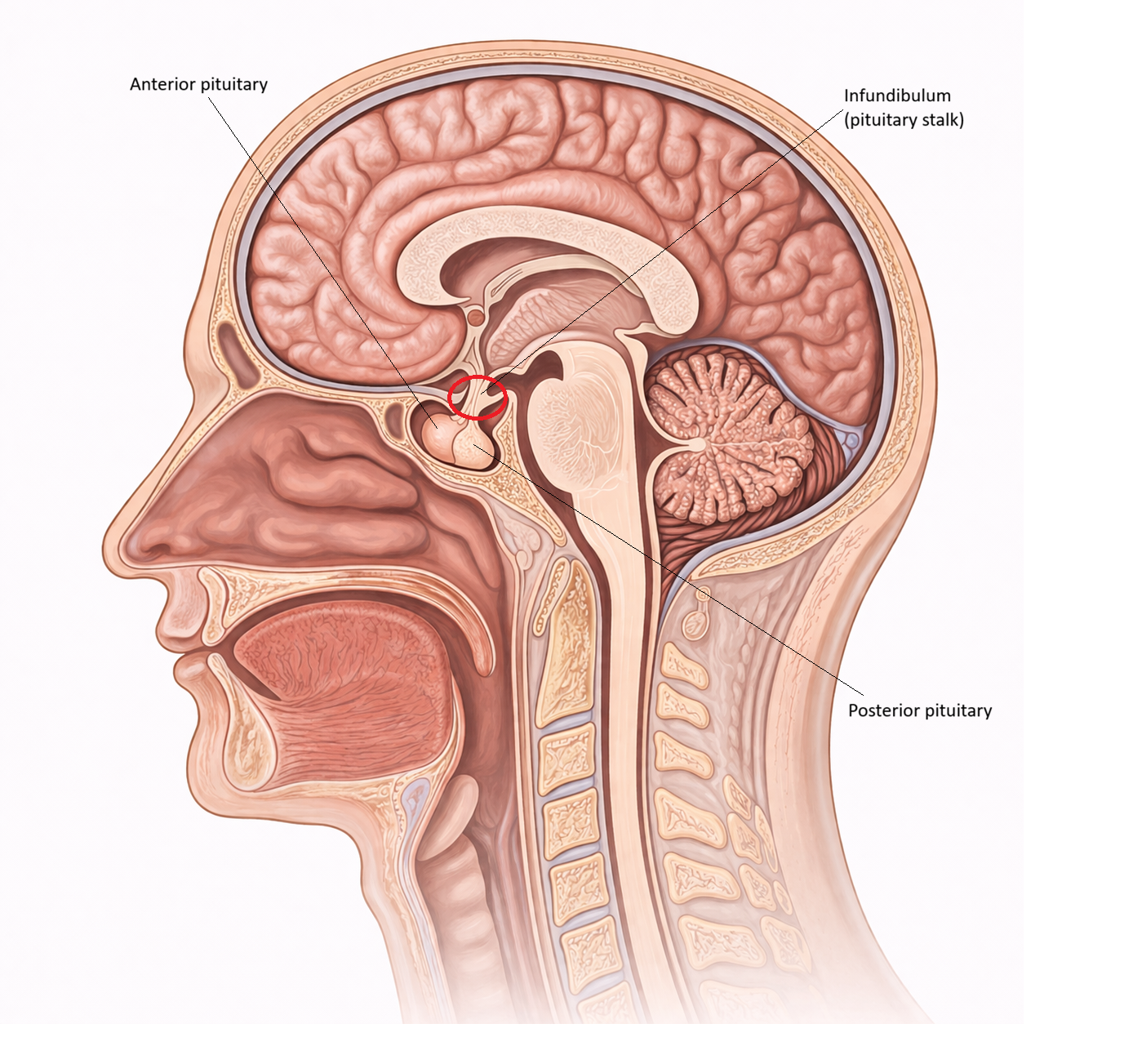
The posterior pituitary, or neurohypophysis, is structurally and functionally distinct from the anterior pituitary. Rather than synthesising hormones itself, it serves as a specialised storage and release site for hormones produced in the hypothalamus. Through tightly coordinated neural regulation, the posterior pituitary releases antidiuretic hormone and oxytocin directly into the bloodstream in response to physiological stimuli. This system allows the brain to exert immediate influence over fluid balance, osmotic regulation, childbirth and lactation. Understanding its structure provides insight into disorders such as diabetes insipidus, syndrome of inappropriate antidiuretic hormone secretion and abnormalities in labour progression.
What You Need to Know
The posterior pituitary is not a true endocrine gland in the same way as the anterior pituitary. It is a specialised neuroendocrine structure that functions as a release site for hormones synthesised in the hypothalamus. Its tissue is composed primarily of unmyelinated axons and nerve terminals originating from neurons in the supraoptic and paraventricular nuclei. These neurons produce antidiuretic hormone (ADH) and oxytocin in their cell bodies, package them into vesicles, and transport them down the hypothalamo-hypophyseal tract for storage in the posterior pituitary.
Unlike anterior pituitary hormones, posterior pituitary hormones do not require releasing hormones or portal circulation for control. Release occurs in response to direct neural stimulation. When specific physiological thresholds are reached—such as increased plasma osmolality or cervical stretch—action potentials travel down hypothalamic neurons, triggering calcium-dependent exocytosis of hormone into the systemic circulation. This arrangement allows rapid, tightly coupled responses to internal changes.
At a functional level, the posterior pituitary plays a critical role in maintaining homeostasis through two hormones with distinct but highly coordinated actions:
Antidiuretic hormone (ADH) increases water reabsorption in the renal collecting ducts, preserving plasma volume and stabilising osmolality in response to dehydration or hypotension
Oxytocin drives uterine contractions during labour and mediates milk ejection during breastfeeding through positive feedback mechanisms initiated by mechanical stimulation
Because hormone release depends on intact neural pathways rather than glandular synthesis, damage to the hypothalamus, pituitary stalk, or axonal transport mechanisms can profoundly impair posterior pituitary function even when the pituitary tissue itself appears structurally normal. This neural dependence explains the characteristic presentation of disorders such as central diabetes insipidus and highlights the posterior pituitary’s unique position at the interface between the nervous and endocrine systems.
Image: The posterior pituitary (neurohypophysis) stores and releases hormones produced by the hypothalamus, primarily ADH and oxytocin.
Beyond the Basics
Neuroanatomical organisation of the posterior pituitary
The posterior pituitary is a specialised neuroendocrine structure composed of unmyelinated axons, pituicytes (specialised glial cells), and an exceptionally rich capillary network. Its axons originate from magnocellular neurons in the supraoptic and paraventricular nuclei of the hypothalamus, forming the hypothalamo-hypophyseal tract. Unlike the anterior pituitary, which synthesises hormones locally, the posterior pituitary primarily functions as a storage and release site for hormones produced in hypothalamic neuronal cell bodies.
Hormones are packaged into neurosecretory vesicles within the hypothalamus and transported down axons via fast axonal transport, a microtubule-dependent system that moves vesicles efficiently over long distances. Within the posterior pituitary, these vesicles accumulate in focal swellings known as Herring bodies. Herring bodies act as local storage depots, allowing the system to release large quantities of hormone quickly when physiological triggers arise. Pituicytes support this arrangement structurally and functionally. By altering their processes and the local extracellular environment, they influence the accessibility of axon terminals to nearby capillaries, helping regulate how readily hormones enter the circulation.
Antidiuretic hormone and physiological control of water balance
Antidiuretic hormone (ADH), also called vasopressin, is essential for maintaining plasma osmolality and circulating volume within a narrow physiological range. Osmoreceptors in the hypothalamus detect subtle rises in plasma osmolality and respond by increasing hypothalamic neuronal firing and ADH release. In parallel, reduced effective circulating volume is detected by baroreceptor pathways, which also stimulate ADH secretion, even when plasma osmolality is not markedly elevated. This dual input explains why ADH can be strongly activated during haemorrhage, shock, or severe dehydration.
In the kidneys, ADH acts predominantly on collecting duct principal cells by binding to V2 receptors and triggering intracellular signalling that increases insertion of aquaporin-2 water channels into the apical membrane. This increases water reabsorption, concentrating urine and conserving free water. At higher concentrations, ADH also binds to V1 receptors on vascular smooth muscle, producing vasoconstriction. This vascular effect becomes clinically relevant when the body is attempting to preserve perfusion pressure during major volume loss, highlighting that ADH is both a water-conserving hormone and, when required, a blood pressure-supporting hormone.
Oxytocin, positive feedback, and coordinated reproductive reflexes
Oxytocin release is most strongly stimulated by mechanical signals, particularly cervical stretch during labour and nipple stimulation during breastfeeding. During labour, stretch receptor input increases hypothalamic neuronal firing, releasing oxytocin into the circulation. Oxytocin then intensifies uterine contractions, which further increases cervical stretch. This forms a positive feedback loop that amplifies contractile activity until delivery occurs. This is physiologically distinct from most endocrine systems, which are governed by negative feedback to stabilise levels rather than escalate them.
In the mammary glands, oxytocin triggers contraction of myoepithelial cells surrounding alveoli and ducts, producing milk ejection rather than milk production. That distinction matters clinically because milk synthesis is largely driven by prolactin, whereas milk let-down depends on oxytocin and is therefore highly sensitive to stress, pain, fatigue, and disrupted neural signalling. Beyond these reproductive roles, oxytocin also acts within the central nervous system. Although its behavioural effects are complex and context-dependent, research suggests oxytocin influences stress modulation, affiliative behaviour, and aspects of emotional processing, reflecting its broader neuromodulatory reach.
Mechanism of hormone release: electrical-to-chemical transduction
Posterior pituitary hormone release is triggered by direct neural activity rather than intermediate endocrine signalling. Action potentials generated in hypothalamic neuron cell bodies propagate down the unmyelinated axons to terminals within the posterior pituitary. When an action potential arrives, it opens voltage-gated calcium channels, and the resulting calcium influx drives exocytosis of hormone-containing vesicles into the extracellular space adjacent to capillaries. Hormones then diffuse into the blood and are carried systemically to their target tissues.
This arrangement enables rapid secretion that is tightly coupled to real-time physiological demands. It also explains why pathology affecting the hypothalamus, pituitary stalk, or axonal transport can cause posterior pituitary hormone deficiency even when the gland itself appears intact on imaging. The posterior pituitary is therefore best understood as a neurosecretory interface, where neural signals are translated into precisely timed endocrine output.
Clinical Connections
Disorders of the posterior pituitary most commonly present through disturbances in water balance, reflecting the central role of antidiuretic hormone in regulating plasma osmolality and circulating volume. Insufficient ADH secretion, or impaired renal responsiveness to ADH, results in diabetes insipidus. In this condition, the kidneys are unable to concentrate urine, leading to the excretion of large volumes of dilute urine and intense thirst. If fluid intake does not match losses, patients rapidly develop dehydration and hypernatraemia. In contrast, excessive ADH secretion produces the syndrome of inappropriate ADH secretion, in which water retention leads to dilutional hyponatraemia, cerebral oedema and neurological symptoms such as confusion, seizures or reduced consciousness.
Posterior pituitary disorders are therefore often recognised through characteristic biochemical and clinical patterns, particularly changes in urine output, serum sodium concentration and plasma osmolality:
Central diabetes insipidus arises from impaired ADH synthesis or release due to hypothalamic or pituitary stalk damage
Nephrogenic diabetes insipidus occurs when the kidneys fail to respond to ADH despite adequate hormone levels
SIADH results from inappropriate, sustained ADH secretion despite normal or low plasma osmolality
Postoperative or traumatic injury to the hypothalamus or pituitary stalk frequently causes transient or permanent ADH dysregulation
Oxytocin-related dysfunction is most clinically apparent in obstetric and postpartum settings. Inadequate oxytocin release can impair the progression of labour by reducing the strength and coordination of uterine contractions, increasing the likelihood of prolonged labour or the need for obstetric intervention. During breastfeeding, insufficient oxytocin activity interferes with milk ejection despite normal milk production, often presenting as poor let-down rather than low supply. Conversely, excessive oxytocin release or inappropriate administration can lead to uterine hyperstimulation, compromising uteroplacental blood flow and increasing fetal distress.
Because the posterior pituitary depends on intact neural connections rather than local hormone synthesis, disorders affecting the hypothalamus, pituitary stalk or axonal transport pathways disproportionately disrupt posterior pituitary function. Trauma, neurosurgery, infiltrative disease and tumours can therefore produce profound hormonal effects even when the gland itself appears structurally normal, underscoring the importance of understanding posterior pituitary physiology when interpreting fluid and electrolyte disturbances in clinical practice.
Concept Check
Why does the posterior pituitary not synthesise its own hormones
How does ADH regulate plasma osmolality and what signals trigger its release
Why is oxytocin secretion considered an example of positive feedback
How do Herring bodies contribute to hormone storage and release
Why do disorders of the pituitary stalk disrupt posterior pituitary function