The Thyroid Gland: Structure, Hormone Synthesis and Metabolic Regulation
The thyroid gland is one of the most metabolically influential organs in the body. Located in the anterior neck and shaped like a butterfly, it produces hormones that regulate basal metabolic rate, thermogenesis, oxygen consumption, growth and the development of the nervous system. Because thyroid hormones act on nearly every cell in the body, even subtle alterations in thyroid function produce widespread physiological changes. The thyroid’s unique follicular structure and iodine-dependent hormone synthesis make it one of the most distinctive glands in the endocrine system.
What You Need to Know
The thyroid gland plays a central role in regulating metabolic activity, growth, and energy balance through the production of thyroid hormones. Its structure and function are closely integrated, with hormone synthesis occurring within specialised follicles and hormonal effects largely determined in peripheral tissues rather than the gland itself.
Several core mechanisms underpin normal thyroid physiology and explain how small changes in hormone levels can have widespread systemic effects:
Thyroxine (T4) is produced in larger quantities than triiodothyronine (T3), but acts primarily as a prohormone, with most biological activity occurring after conversion to T3 in peripheral tissues.
Peripheral conversion of T4 to T3 is mediated by deiodinase enzymes, allowing individual tissues to locally regulate thyroid hormone activity according to metabolic demand.
T3 increases metabolic rate by upregulating gene transcription involved in protein synthesis, mitochondrial activity, and thermogenesis, thereby increasing oxygen consumption and heat production.
Thyroid-stimulating hormone (TSH) from the anterior pituitary regulates thyroid hormone synthesis and release through negative feedback, maintaining relatively stable circulating hormone levels.
Adequate iodine intake is essential for hormone synthesis, as iodine is a critical structural component of both T3 and T4.
In addition to follicular cells, the thyroid contains parafollicular (C) cells that secrete calcitonin, which contributes to calcium homeostasis by opposing bone resorption. While calcitonin plays a relatively minor role in day-to-day calcium regulation compared with parathyroid hormone, its presence highlights the thyroid’s involvement beyond metabolic control.Normal thyroid function depends not only on hormone production within the gland but also on effective peripheral conversion, intact feedback regulation, and sufficient substrate availability.
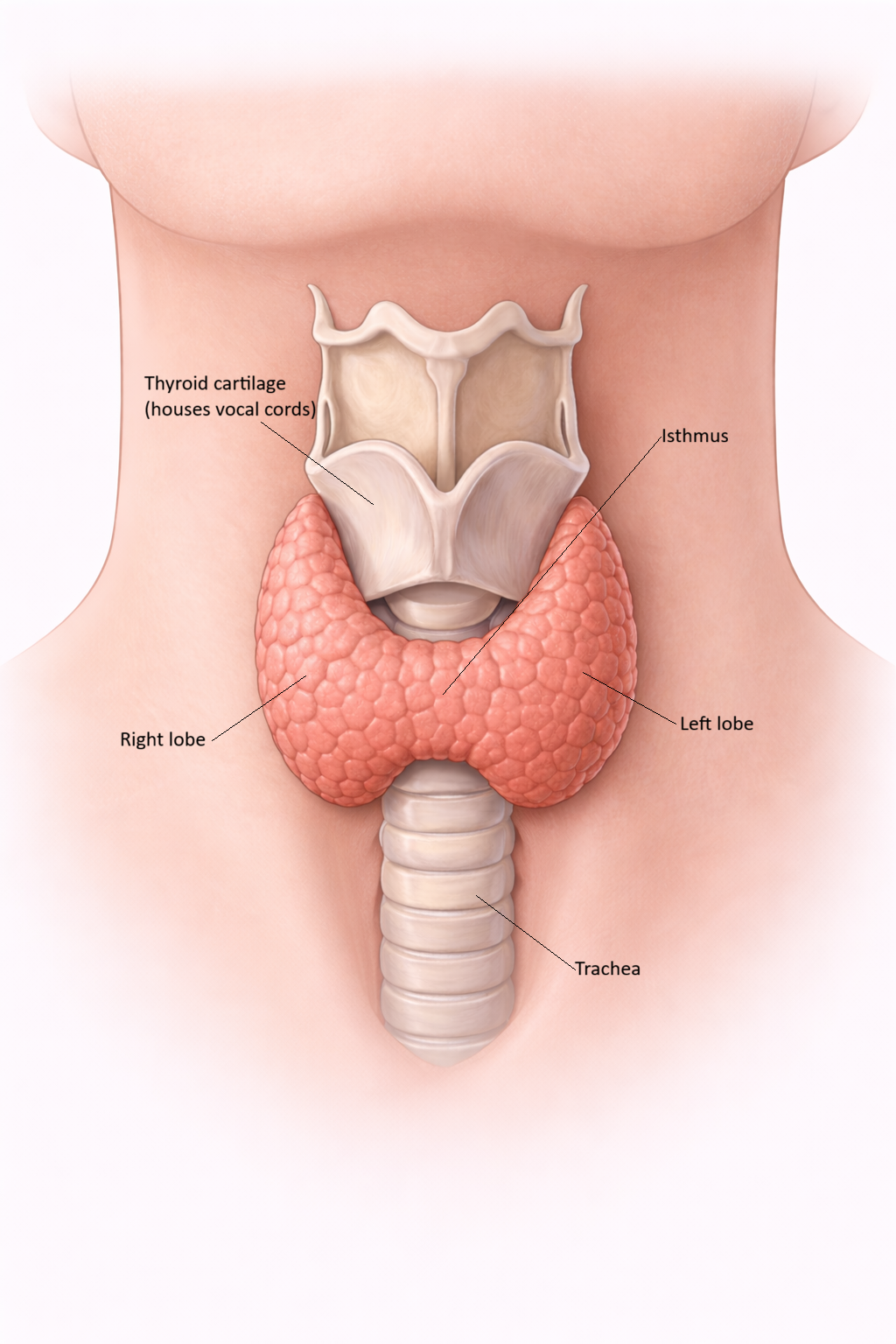
Image: The thyroid gland sits in the anterior neck, wrapping around the upper trachea. Its two lobes are connected by an isthmus, and it functions to regulate metabolism through hormone secretion.
Beyond the Basics
Follicular architecture and hormone synthesis
The functional unit of the thyroid gland is the follicle, a highly specialised spherical structure designed to support both hormone synthesis and long-term storage. Each follicle is lined by a single layer of follicular epithelial cells that surround a central lumen filled with colloid, a viscous protein-rich substance composed primarily of thyroglobulin. This arrangement is unique among endocrine glands, as thyroid hormones are synthesised extracellularly within the colloid and stored in large quantities before release, allowing the body to maintain hormone availability over extended periods.
Thyroid hormone synthesis begins with active iodide uptake from the circulation into follicular cells via sodium–iodide symporters, a process that is strongly stimulated by thyroid-stimulating hormone. Iodide is transported into the follicular lumen, where it is oxidised and attached to tyrosine residues on thyroglobulin in a process known as organification. This leads to the formation of monoiodotyrosine and diiodotyrosine, which subsequently undergo coupling reactions to generate triiodothyronine and thyroxine. These hormonally active molecules remain bound to thyroglobulin within the colloid, forming a large extracellular hormone reserve that can be rapidly mobilised when required.
Hormone release, transport, and peripheral activation
When circulating thyroid hormone levels fall, follicular cells respond by endocytosing colloid droplets from the follicular lumen. Within the cell, lysosomal enzymes cleave thyroglobulin, releasing free T3 and T4, which then diffuse into the bloodstream. The majority of released hormone binds to plasma proteins, particularly thyroxine-binding globulin, which serves to stabilise circulating hormone levels and extend hormone half-life. Only a small unbound fraction remains biologically active, but this free component is tightly regulated and reflects true hormonal activity at the tissue level.
Although both T3 and T4 are secreted, T4 predominates in circulation and functions largely as a prohormone. Target tissues express deiodinase enzymes that convert T4 into either active T3 or inactive reverse T3, enabling local control of thyroid hormone action. This peripheral regulation allows tissues with high metabolic demand, such as the brain, liver, and skeletal muscle, to fine-tune intracellular hormone availability independent of circulating concentrations. As a result, thyroid hormone action is determined not only by glandular output but also by tissue-specific enzymatic activity.
Vascular and lymphatic integration
The thyroid gland is among the most highly vascularised organs in the body, a feature that reflects its role in rapid hormone exchange and iodine delivery. Rich arterial supply ensures efficient uptake of iodide from the circulation and enables swift distribution of newly released hormones to target tissues. This extensive blood flow also explains why changes in thyroid hormone production can have rapid and widespread systemic effects.
In parallel, the lymphatic drainage of the thyroid has important clinical implications. Lymphatic channels connect the gland to central and lateral cervical lymph nodes, providing potential pathways for the spread of malignant cells. Understanding this structural organisation is essential for interpreting imaging findings and planning surgical intervention in thyroid disease, particularly in the context of malignancy.
Calcitonin and parafollicular cell function
In addition to follicular cells, the thyroid contains parafollicular, or C cells, which arise from a different embryological origin and serve a distinct endocrine function. These cells secrete calcitonin, a hormone that lowers blood calcium levels by inhibiting osteoclast-mediated bone resorption. In humans, calcitonin plays a relatively minor role in day-to-day calcium regulation compared with parathyroid hormone and vitamin D.
Despite its limited physiological role, calcitonin has significant clinical relevance. Excessive calcitonin production is a hallmark of medullary thyroid carcinoma, making it a valuable diagnostic and monitoring marker. This illustrates how specialised thyroid cell populations can have disproportionate importance in disease states, even when their normal physiological contribution is modest.
Feedback regulation and metabolic stability
Thyroid hormone production is tightly controlled by the hypothalamic–pituitary–thyroid axis, which maintains metabolic stability through dynamic feedback mechanisms. Thyrotropin-releasing hormone from the hypothalamus stimulates thyroid-stimulating hormone release from the anterior pituitary, which in turn promotes iodine uptake, hormone synthesis, and follicular cell activity within the thyroid gland. Rising levels of circulating T3 and T4 exert negative feedback at both hypothalamic and pituitary levels, suppressing further hormone production.
This regulatory system allows the thyroid axis to respond to physiological demands such as cold exposure, changes in nutritional status, and stress. Rather than operating as a simple on–off mechanism, feedback regulation continuously adjusts hormone output to maintain homeostasis. As a result, thyroid function reflects an integrated balance between central control, glandular capacity, and peripheral hormone activation.
Clinical Connections
Thyroid dysfunction shows up often in everyday clinical settings and is rarely labelled as “thyroid” at first. People usually present with vague, slowly developing symptoms, or thyroid abnormalities are picked up incidentally on routine blood tests. Because thyroid hormones affect metabolism, heart rate, temperature regulation, and mental processing, problems with thyroid function tend to affect several systems at once.
In clinical practice, thyroid-related presentations often follow a few recognisable patterns:
Increased thyroid hormone activity driving a faster metabolic state
Reduced thyroid hormone activity leading to overall metabolic slowing
Structural changes in the thyroid detected on examination or imaging
When thyroid hormone levels are high, the body is effectively running faster than normal. People may have a persistently elevated heart rate, feel hot when others do not, lose weight without trying, or appear restless and shaky. These features are often first noticed during routine observations or when symptoms seem out of proportion to the situation, such as ongoing tachycardia without an obvious cause.
When thyroid hormone levels are low, everything slows down. Fatigue, cold intolerance, weight gain, constipation, slowed heart rate, and poor concentration are common, but they often develop gradually. Because these changes can be subtle, they are frequently attributed to stress, ageing, or other chronic conditions unless thyroid function is specifically considered.
Structural thyroid problems are also commonly encountered. An enlarged thyroid may be noticed on inspection or palpation, while nodules are often found incidentally on imaging done for other reasons. Autoimmune thyroid disease can change both hormone levels and the physical structure of the gland over time.
Concept Check
Why is iodine essential for thyroid hormone synthesis
How does the follicular architecture of the thyroid support both hormone storage and production
Why is T3 more biologically active than T4
How does the hypothalamic–pituitary–thyroid axis maintain stable hormone levels
What structural features of the thyroid explain its susceptibility to autoimmune and nodular diseases