WOUND HEALING — Stage 4: Remodelling (Maturation)
The remodelling or maturation phase is the final stage of wound healing, beginning around 3 weeks after injury and continuing for months to years. Whereas the proliferative phase focuses on filling the wound and restoring coverage, remodelling transforms fragile, newly formed tissue into a stronger and more structurally organised scar. This stage involves the extensive reorganisation of collagen fibres, reduction in cellularity and vascularity, and refinement of the extracellular matrix. Although the wound gradually becomes stronger, it never fully regains the tensile strength of uninjured skin. The remodelling phase is therefore essential not only for restoring functional integrity but also for determining long-term cosmetic and biomechanical outcomes.
What You Need to Know
Remodelling is the final and longest phase of wound healing, beginning once the wound surface is closed and granulation tissue has filled the defect. During this stage, the temporary tissue formed in proliferation is gradually reorganised into stronger, more functional tissue. Although the wound may appear healed externally, extensive cellular and structural changes continue beneath the surface for months to years.
Key processes that define remodelling include:
Replacement of type III collagen with stronger type I collagen
Reorientation of collagen fibres along lines of mechanical tension
Gradual reduction in fibroblast and myofibroblast numbers through apoptosis
Regression of excess capillaries and reduction in vascularity
As collagen is remodelled and realigned, tensile strength steadily increases. Fibres become thicker, more tightly packed and better organised, allowing the tissue to tolerate everyday mechanical stress. At the same time, unnecessary blood vessels are pruned, reducing metabolic demand and giving the scar its flatter, paler appearance. Despite these improvements, healed tissue never fully regains original strength, typically reaching only about 70 to 80 percent of uninjured skin. Disruption of collagen balance or prolonged mechanical tension during this phase can shift remodelling toward abnormal scarring, making this stage critical for determining long-term structure and function.
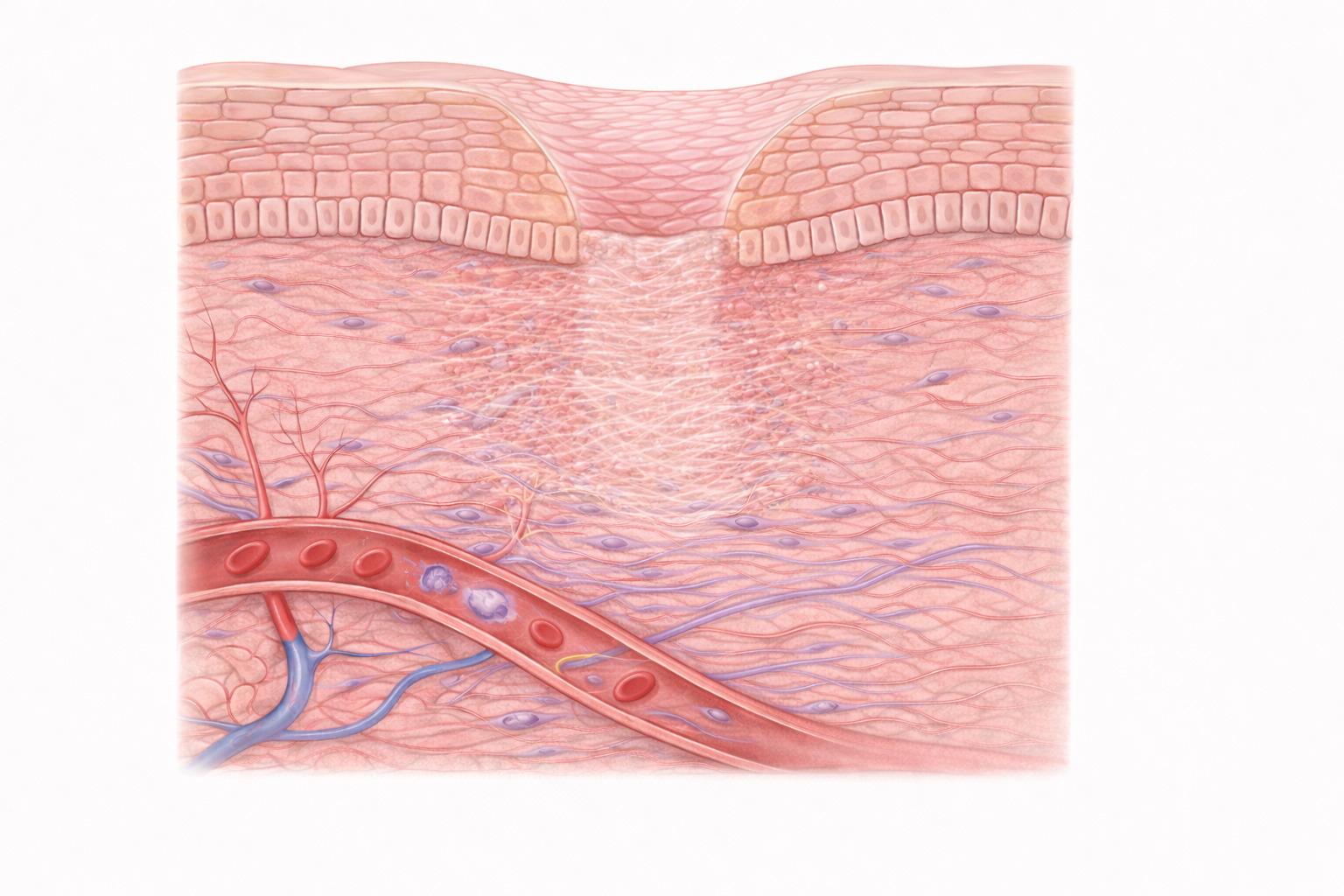
Image: Collagen fibres are reorganised and strengthened, forming a more structured scar as cellular activity and vascularity decrease.
Beyond the Basics
Transition from type III to type I collagen
The defining event of the remodelling phase is the gradual replacement of type III collagen with type I collagen. Type III collagen, produced during proliferation, provides early flexibility and volume but offers limited tensile strength. As healing progresses, fibroblasts increasingly synthesise type I collagen under the influence of transforming growth factor beta. This collagen forms thicker, stronger fibrils capable of withstanding mechanical load.
At the same time, matrix metalloproteinases selectively degrade type III collagen and other provisional extracellular matrix components. Their activity is tightly regulated by tissue inhibitors of metalloproteinases, ensuring degradation and synthesis remain balanced. This controlled turnover allows older, weaker collagen to be removed and replaced with fibres that are better organised and more mechanically efficient, rather than simply increasing collagen quantity.
Collagen realignment and mechanical adaptation
Early collagen deposition follows a disorganised, basket-weave pattern that provides bulk but limited strength. During remodelling, fibroblasts respond to mechanical forces acting on the wound and gradually realign collagen fibres along lines of tension. This reorientation significantly increases tensile strength without requiring large increases in collagen mass.
Cross-linking between collagen molecules further stiffens and stabilises the matrix, making the tissue progressively more durable. Because collagen organisation responds to mechanical load, the forces applied to a healing wound influence scar quality. Controlled movement, gradual loading and appropriate support can therefore shape fibre alignment, while excessive tension or repeated trauma may disrupt organisation and worsen scarring.
Reduction in cellularity and vascularity
Granulation tissue formed during proliferation is highly cellular and richly vascular to support rapid repair. As remodelling begins, much of this cellular and vascular infrastructure is no longer required. Fibroblast numbers decline once collagen deposition stabilises, and myofibroblasts undergo apoptosis as wound contraction completes. Simultaneously, excess capillaries regress through programmed vessel pruning.
These changes reduce metabolic demand and alter the appearance of the wound. As vascular density decreases, the scar becomes paler and flatter, and tissue hydration falls as the matrix becomes more collagen-dense. This shift reflects the transition from active regeneration to long-term structural consolidation.
Progressive strengthening and functional limits
Tensile strength increases rapidly during the early weeks of remodelling, then more slowly over subsequent months. Final strength depends on the balance between collagen synthesis, degradation and alignment. Although fibres become thicker and more organised, the repaired tissue never fully recreates the complex architecture of uninjured dermis, which includes elastin networks, adnexal structures and specialised extracellular matrix components.
As a result, even well-remodelled scars achieve only around 70 to 80 percent of original tissue strength. This persistent vulnerability explains why newly healed wounds remain susceptible to breakdown under excessive mechanical stress and require ongoing protection.
Pathological scarring and remodelling failure
When remodelling becomes dysregulated, abnormal scarring can develop. Excessive or prolonged fibroblast activity leads to continued collagen deposition and raised scars. Hypertrophic scars remain confined to the wound margins but are thick, firm and often erythematous due to persistent vascularity. Keloids represent a more exaggerated response, extending beyond the original wound boundary and reflecting profound dysregulation of collagen synthesis and degradation.
In contrast, insufficient collagen production or poor organisation results in weak, atrophic scars. These are more likely in settings of malnutrition, chronic inflammation, prolonged corticosteroid exposure or systemic illness, and are prone to breakdown. The remodelling phase therefore plays a decisive role in determining both the mechanical integrity and long-term appearance of healed tissue.
Clinical Connections
Outcomes in the remodelling phase are strongly influenced by what occurred earlier in healing. Poor perfusion, infection, prolonged inflammation or repeated trauma during the inflammatory and proliferative stages lead to collagen that is poorly organised and mechanically weak. By the time remodelling begins, these earlier disruptions cannot be undone, but their consequences can be moderated through careful scar management and controlled mechanical loading.
In practice, remodelling problems most often present as:
Excessive collagen deposition resulting in raised or rigid scars
Poor collagen organisation leading to weak, widened or unstable scars
Excessive tension causing contracture formation, particularly near joints
Therapeutic strategies during this phase aim to influence collagen behaviour rather than accelerate healing. Silicone gel sheeting and pressure therapy help flatten scars by altering hydration, oxygen tension and fibroblast activity. Corticosteroid injections suppress excessive collagen synthesis in hypertrophic scars and keloids, while laser therapies target vascularity and collagen remodelling to improve texture and pliability. These interventions are most effective when introduced early in the remodelling phase, before collagen architecture becomes fixed.
Mechanical forces play a major role in determining final scar structure. Wounds across joints or in areas of high tension are particularly vulnerable to widening and contracture if movement is uncontrolled or excessive. In burn injuries, where collagen deposition is often exaggerated, aggressive contracture prevention through splinting, positioning and physiotherapy is essential to preserve range of motion and function. Understanding the timing and progression of remodelling allows clinicians to guide rehabilitation, select appropriate scar interventions and set realistic expectations for long-term appearance and strength.
Concept Check
Why is the transition from type III to type I collagen essential for restoring wound strength?
How do mechanical forces influence collagen orientation during remodelling?
Why does a healed wound never regain full tensile strength?
What differentiates a hypertrophic scar from a keloid in terms of pathophysiology?
How do MMPs and TIMPs contribute to collagen reorganisation during maturation?